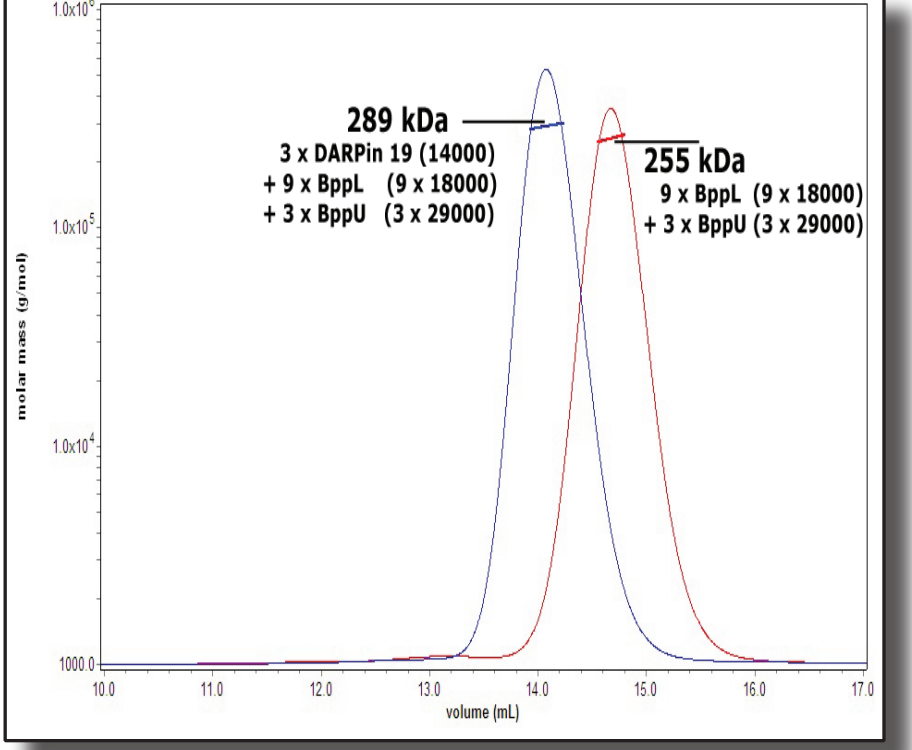
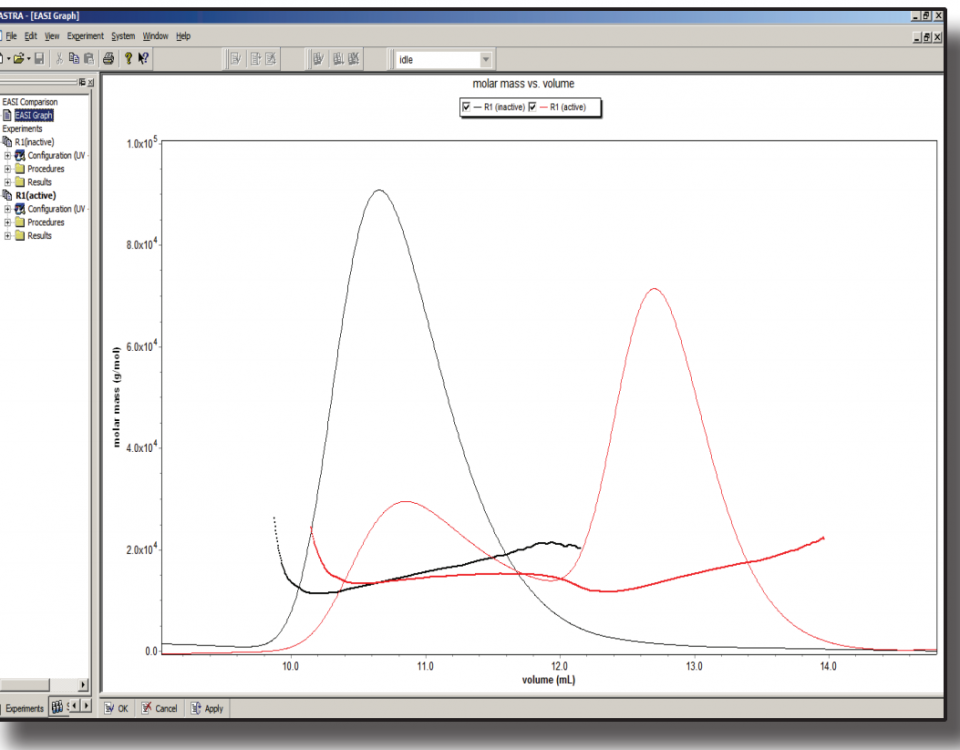
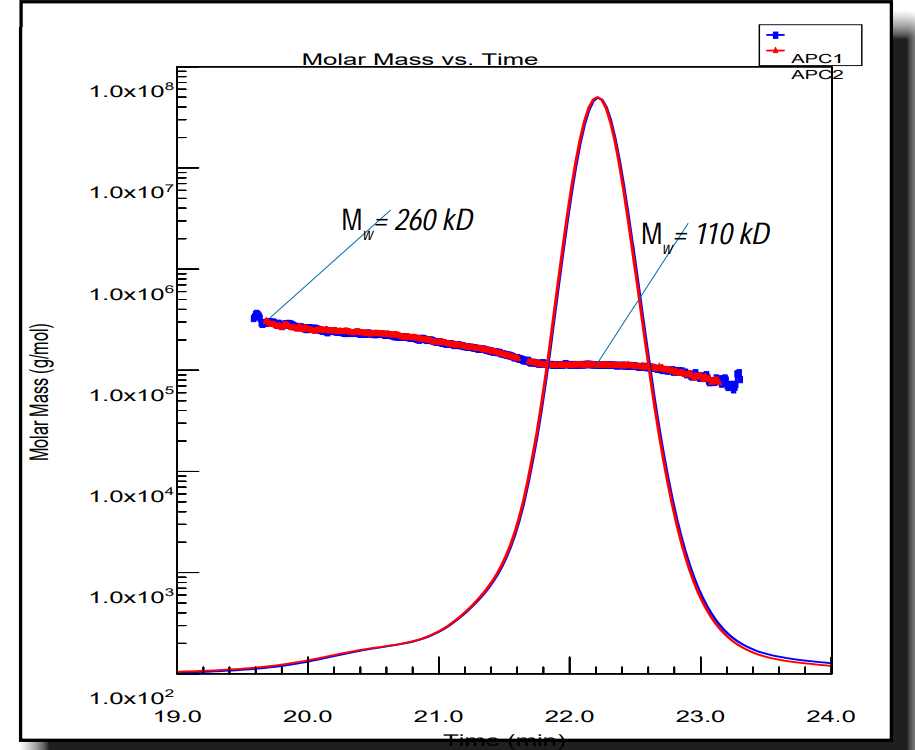
In this application note, we selected three Designed Ankyrin Repeat Proteins (DARPins) and binded to the baseplate of the TP901-1 lactococcal phage. As a part of a structure/function study, we characterized both mass and stoichiometry of each DARPin in complex with either the RBP alone or a multi-protein complex, constituting part of the TP901-1 baseplate and formed by the RBP and the BppU protein.
Bacteria use two-component signal transduction pathways, which comprise a histidine kinase (HK) and a response regulator protein (RR). Extracellular stimuli sensed by the HK induce an autophosphorylation reaction at a histidine residue of the enzyme that is subsequently transferred to a specific aspartate residue at the receiver domain of a cognate RR.
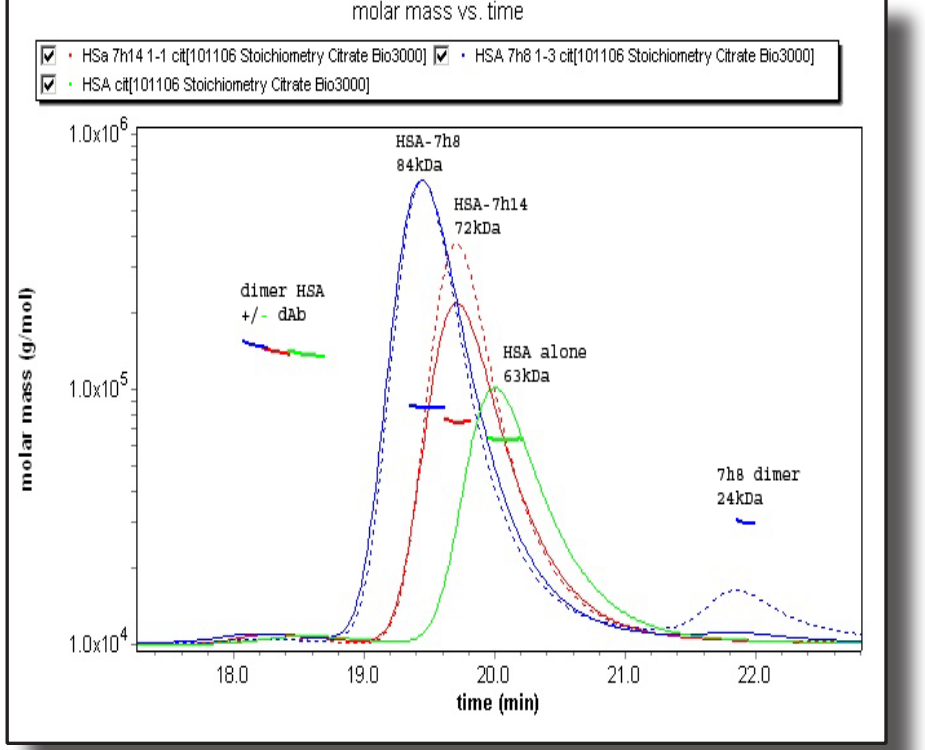
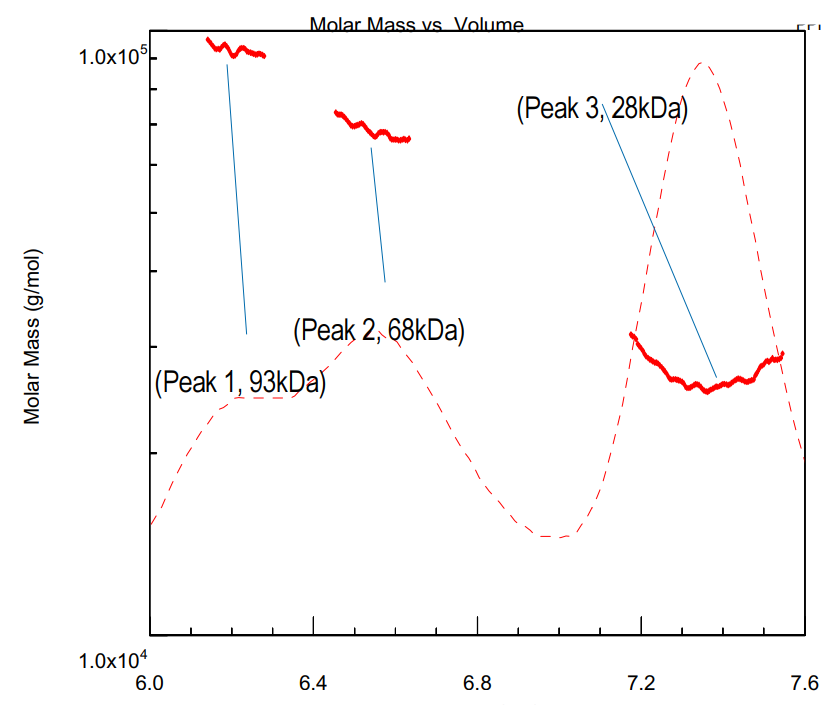
Domain antibodies (dAbs, 12-14kDa) are small molecules comprising Vh or Vk domains of human IgG molecules, which are capable of binding to various antigens. In this application note, we assess the stoichiometry of interaction between dAbs and their corresponding antigen–in this instance, human serum albumin (HSA), using SEC MALS.
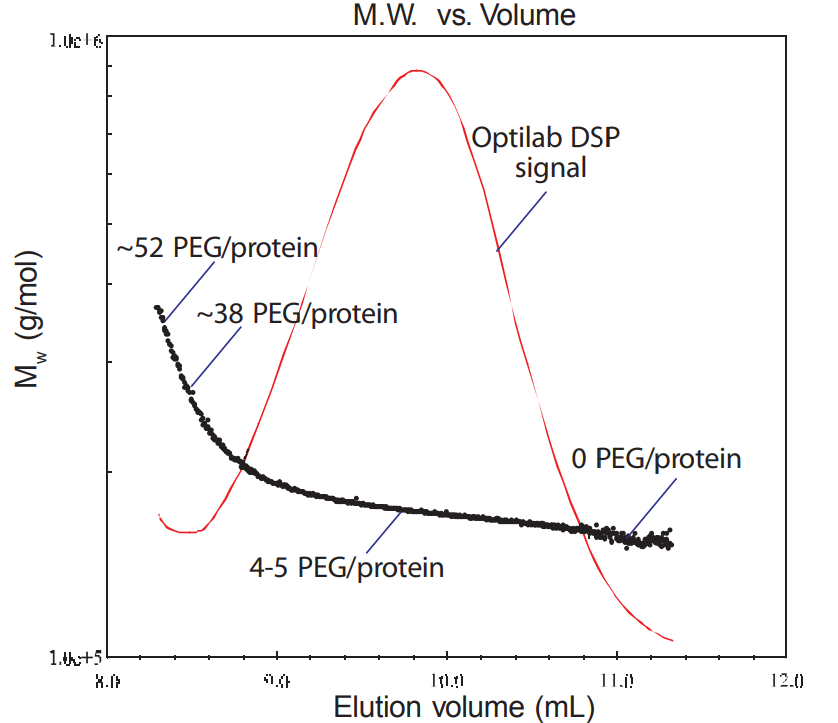
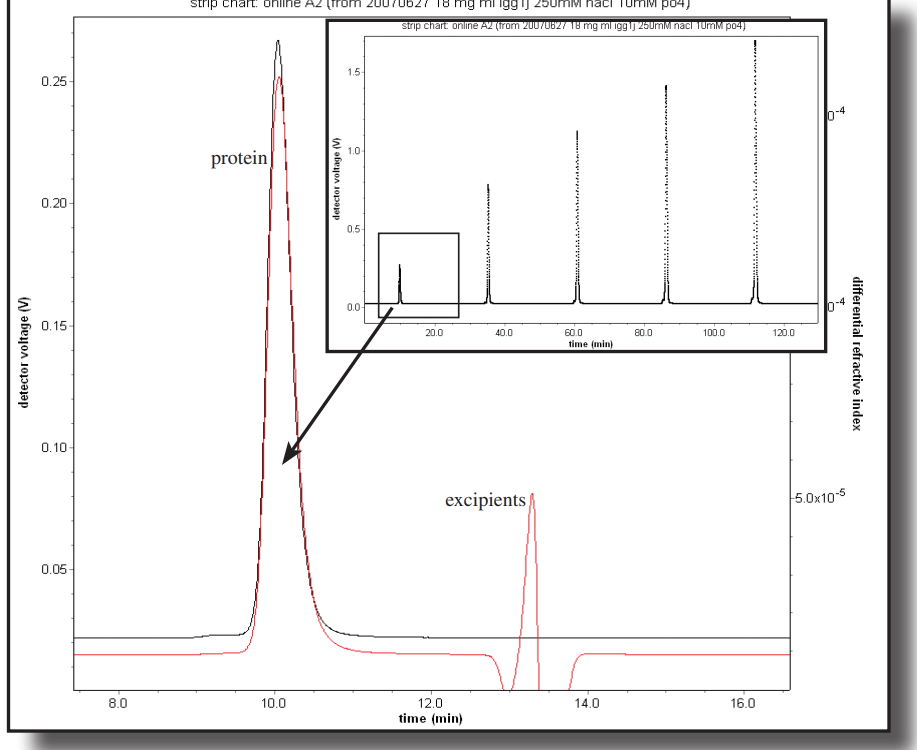
The rise of biotechnology has spawned different means of delivering drugs to the human body.Among the many problems encountered is the rapid rejection or clearance of a protein before it has accomplished its task. In this application note, a monoclonal antibody was measured before and after PEGylation. Learn more.
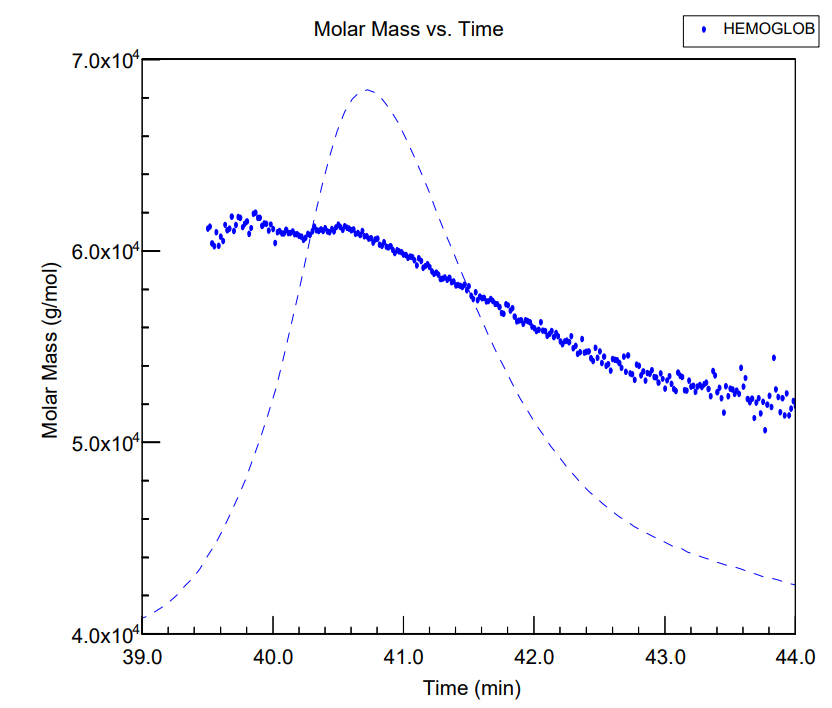
When oxygen is released, carbon dioxide binds to hemoglobin to be transported out of the body. Human hemoglobin contains four polypeptides, in which the tetramer equilibrates with a dimer form, depending on the conditions. Using size-exclusion chromatography (SEC) in combination with multi-angle light scattering (MALS), a miniDAWN detector and Optilab DSP interferometric refractometer were used for the characterization.
Cellular proteins that are part of the protein synthesis machinery are often up-regulated under such conditions. Large quantities of the recombinant protein can be bound to these cellular proteins, making purification difficult. Analytical size exclusion chromatography, followed by on-line direct mass detection by multi-angle light scattering (MALS), showed that a significant amount of the recombinant protein was not bound to the rotamase.
When using MALS detectors to characterize polymers that have chromophores, corrections are needed to compensate for the absorption and/or fluorescence at the operating wavelength in order to obtain accurate molar mass determinations.
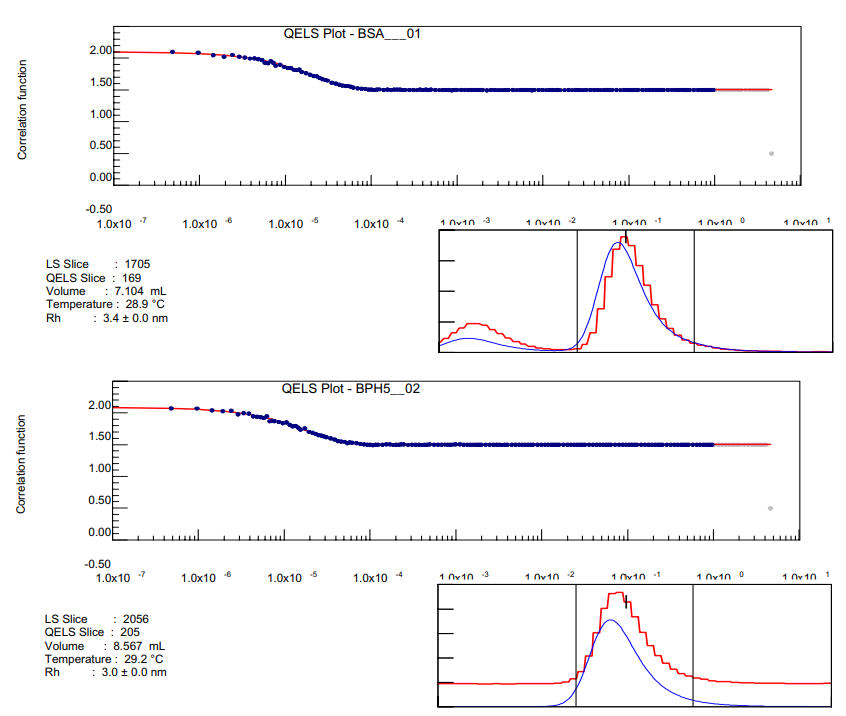
On-line quasi-elastic light scattering (QELS) together with MALS detection provides insight into the structure of polymers when both hydrodynamic and root-mean square radii are measured. In this application note, we report how the on-line WyattQELS detector helps to evaluate the quality of the SEC separation and estimate the shape of proteins.
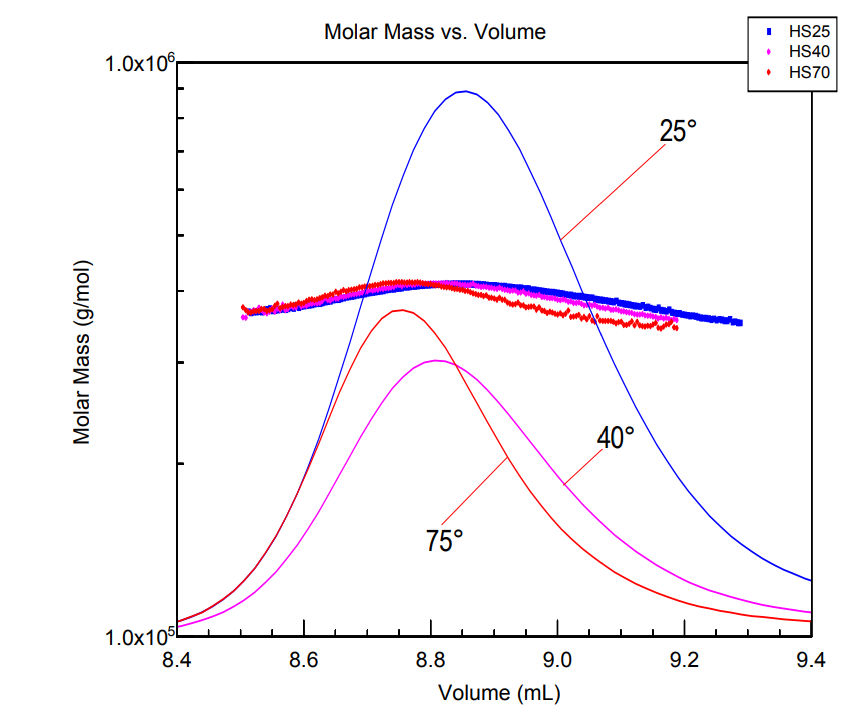
In response to stresses, including heat, oxidants and toxins, all cells produce a common set of heat shock proteins (HSPs). There are four classes of high molar mass HSPs and one family of small HSPs (sHSPs). The sHSPs (12-43 kDa) tend to form large multimeric complexes. Size exclusion chromatography (SEC) has often been used to characterize these complexes.
The sarcoplasmic reticulum (SR) plays an essential role in muscle excitation-contraction coupling by regulating the cytosolic free Ca2+ concentration. In order to study the plausible coupling between the Ca2+ binding and the polymerization of CSQs, we have for the first time employed Multi-Angle Light Scattering (MALS) for the exact measurement of Ca2+ dependent polymeric state of CSQs.
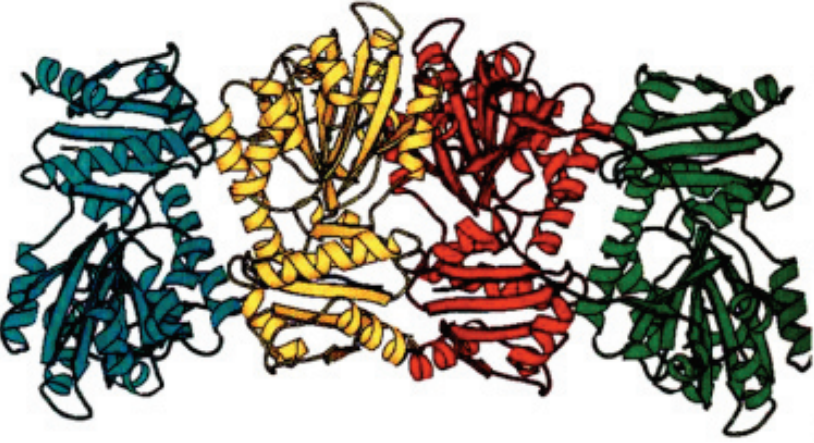
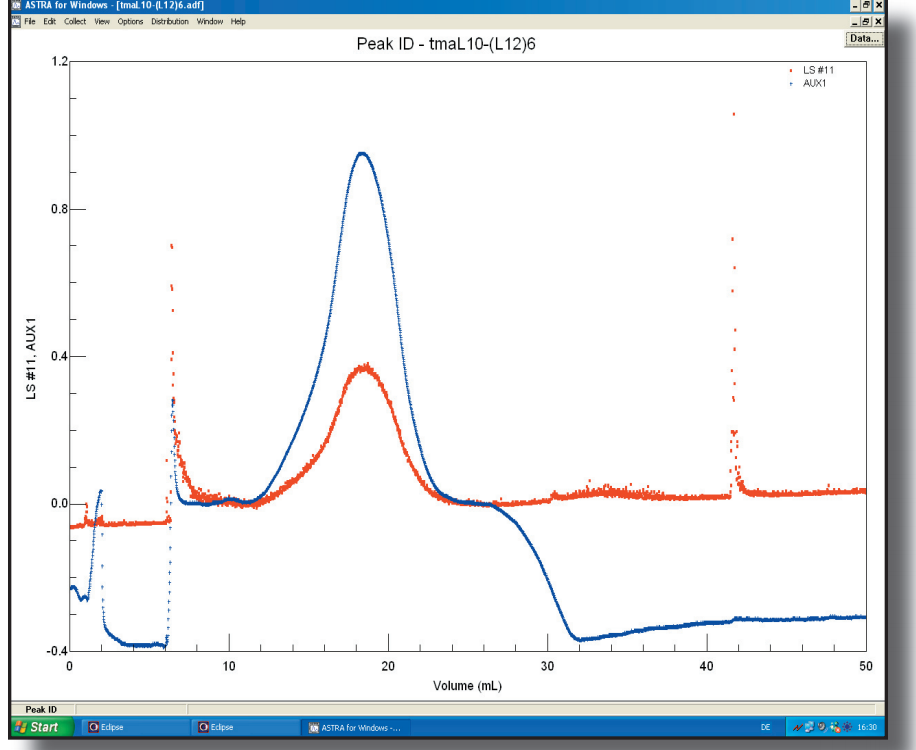
To elucidate the mechanism of action of the L7/L12 stalk of the large ribosomal subunit on the ribosome, we have determined the crystal structure of the protein L10 in complex with L12 Nterminal domain (NTD) from the hyperthermophilic bacterium Thermotoga maritima. Read more here.
Measurement of weak protein-solvent interactions is essential in a wide array of processes, such as stabilization of therapeutic protein formulations, purification of protein mixtures, and crystallization of proteins. Wyatt Technology has developed an accurate, automated method for measuring weak protein-solvent interactions. Read more about it here.