AAV characterization and quantitation
Adeno-associated viruses (AAVs) have emerged as one of the main delivery vehicles for gene therapies. With their rapid growth in development and production, it is essential to implement simple, fast AAV quantification methods for accurately determining the critical quality attributes (CQA) of AAV serotypes, as well as providing extended characterization. Analytical methods based on dynamic light scattering (DLS) and multi-angle light scattering (MALS) are advantageous for quantifying CQAs and other characteristics of AAV vectors: they require no special reagents, extensive sample preparation or calibration curves, are highly reproducible, and results are obtained in just seconds to minutes.
Click on the buttons to explore how light scattering is used to measure different properties of viral vectors or continue reading to learn about Wyatt solutions to industry and regulatory needs for AAVs.
AAV analytical methods based on light scattering
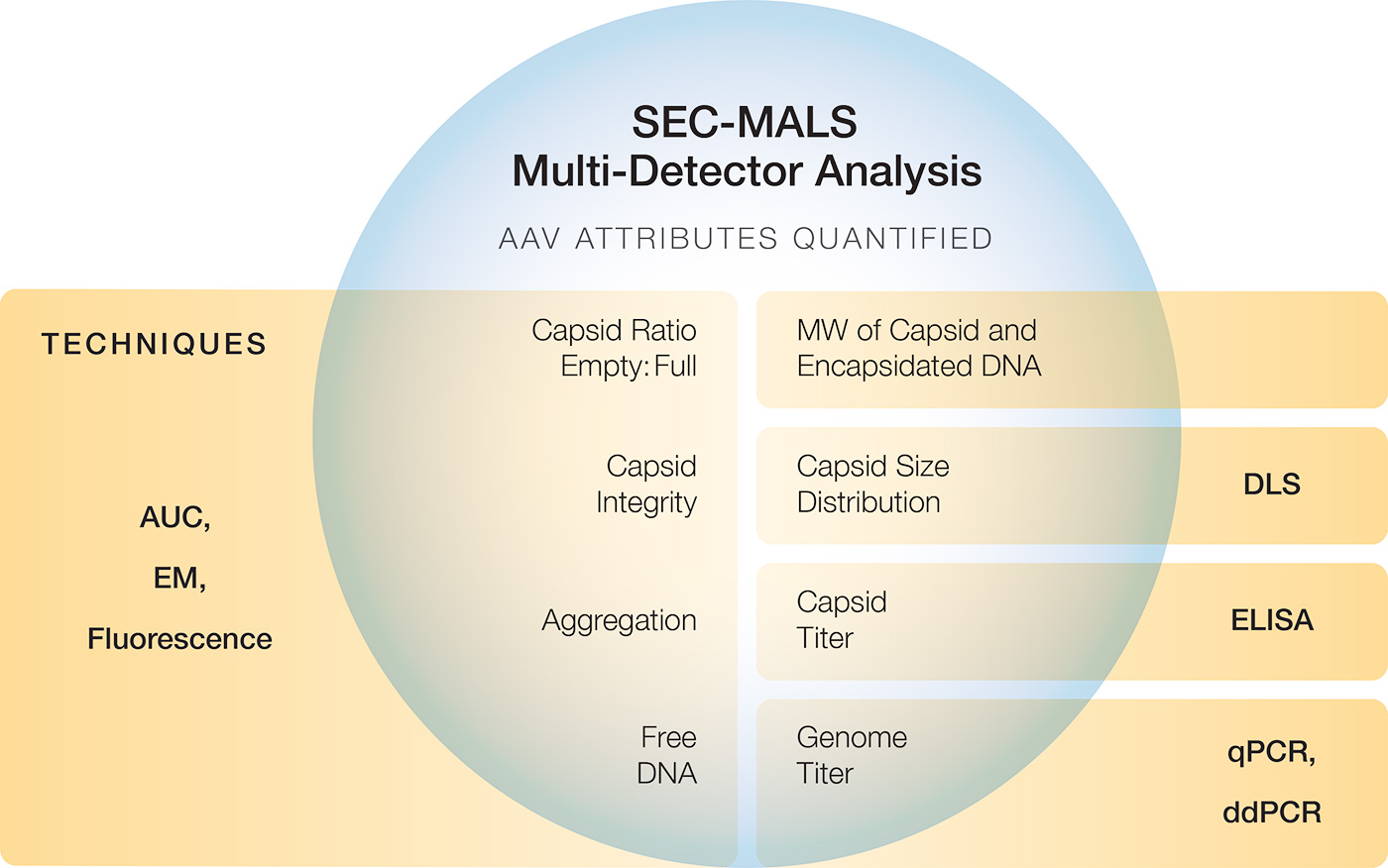
SEC-MALS: Multiple AAV CQAs
Size-exclusion chromatography combined with multi-angle light scattering (SEC-MALS) delivers high-precision AAV quantitation in a single 30-minute experiment. The following AAV quality attributes are measured simultaneously by SEC-MALS:
The same experiments can provide extended characterization of AAV vectors:
- Capsid integrity
- Capsid size (Rh or Rg) and distribution
- Capsid and genome molar mass (Mw)
- Impurities such as free DNA or host-cell proteins
Instrument control, data acquisition, analysis and reporting are all provided by ASTRA™ software with the Viral Vector Analysis module. We provide a platform SEC-MALS method, specifically built for AAV quantitation and is easily customizable for every product and serotype. With ASTRA’s 21 CFR Part 11-compliant option, SEC-MALS is readily positioned for supporting IND or BLA analytics packages, lot-to-lot comparisons and release assays of AAV-based gene therapy products.
The white paper WP1615: SEC-MALS for absolute biophysical characterization discusses SEC- MALS technology for biophysical characterization of several biomolecules.
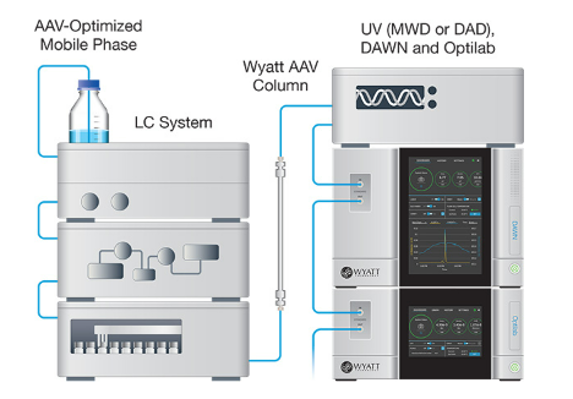
A complete SEC-MALS system includes Wyatt’s DAWN MALS detector, an Optilab dRI detector, an AAV separation column and an industry-standard HPLC pump, an autosampler, and a UV detector.
AAV SOP guidance manual
This comprehensive document serves as a complete reference and pathway to customizing, qualifying and implementing the SEC-MALS AAV method for your specific needs.
AAV method implementation & training
Wyatt has developed a personalized AAV training program that saves significant time and effort in learning, method development and validation.
DLS: High-throughput for formulation screening and process characterization
DLS provides rapid, low-volume, non-destructive AAV quantification to determine size, size distribution and physical titer. Typical experiment times are 30 seconds per sample.
AAV characterization by DLS is essential for:
- Monitoring titer of fractions during AAV production, especially in downstream processes
- Screening buffers and serotypes for colloidal stability and propensity to aggregate
- Assessing thermal stability by measuring the aggregation onset temperature, melting temperature, or the temperature dependence of aggregation kinetics
- Identifying SEC mobile phase or formulation buffers that promote well-assembled product
The DynaPro™ Plate Reader is the premier tool for high-throughput DLS: it evaluates product quality, quantity and stability for up to over a thousand samples at a time. Since the DynaPro Plate Reader measures directly in standard microwell plates, consumable and labor costs are minimal, and common automation tools such as robotic pipettors may be incorporated in the workflow.
The white paper WP5003: Automated dynamic and static light scattering in microwell plates for fast, productive development of biologics describes the capabilities of the DynaPro Plate Reader for performing multiple assays.
The DynaPro™ NanoStar™ performs the same measurements in quartz cuvettes with volumes as low as 2 µL and in disposable cuvettes with just 4 µL.
With the suite of instrument control, analyses and visualization tools provided by DYNAMICS™ software, scientists can easily evaluate multiple product attributes over many samples, to develop stable, robust products and processes.
DynaPro® Plate Reader
High-throughput, automated measurements.

DynaPro™ NanoStar™
Microcuvette-based, walk-up measurements.

A complete FFF-MALS system includes Wyatt’s Eclipse, DAWN MALS detector, Optilab dRI detector, a separation channel and an industry-standard HPLC pump, an autosampler, and a UV detector.
FFF-MALS: Extended aggregate characterization
For the characterization of large AAV aggregates or other large species, field-flow fractionation in combination with MALS (FFF-MALS) is the method of choice. The Eclipse™ FFF system separates particles from 1 – 1000 nm in an open channel with no stationary phase and is invaluable for analyzing particles that are too large for separation by SEC or may be removed or dissociated by column shear.
For more information on the theory and applications of FFF-MALS, please refer to our FFF-MALS page, or read AN2004 which describes the characterization and quantitation of large AAV aggregates using FFF-MALS.
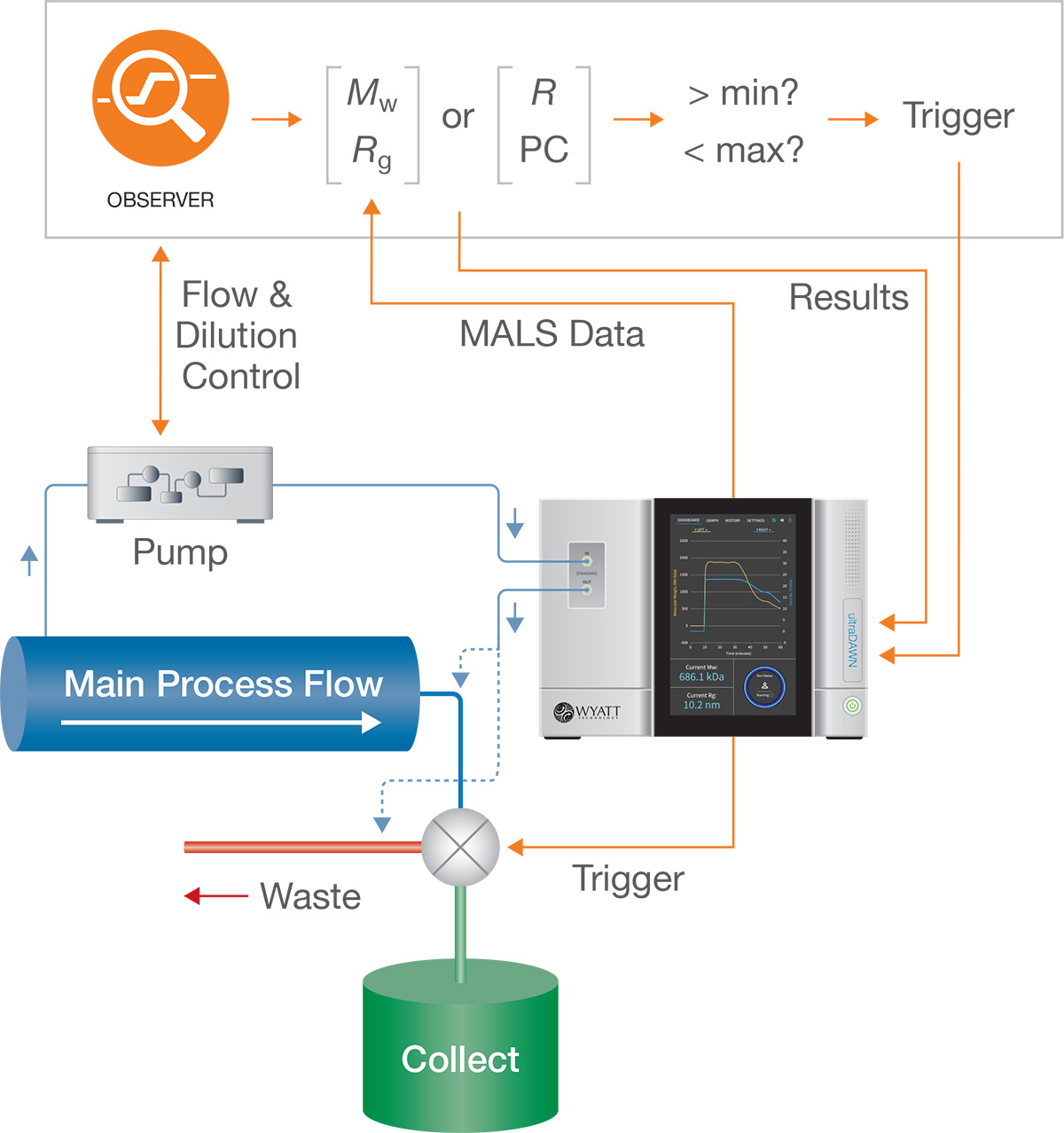
RT-MALS: Process analytical technology (PAT) for AAVs
RT-MALS is a real-time technology for determining empty/full ratio, titer, molar mass and radius. It is valuable in both early- and late stage process environments to monitor quality, collect the appropriate fraction, or flag deviations from acceptable AAV attribute values. RT-MALS is implemented with an ultraDAWN™ MALS instrument and OBSERVER™ software.
Benefits of RT-MALS:
- Accelerate process development and scale-up
- Increase yield, reduce overall time until lot release
- Maintain product quality with confidence
Applications of RT-MALS:
- Chromatographic purification and enrichment of full capsids
- Concentration of viral vectors by ultrafiltration
Learn how RT-MALS is used to accelerate the development of a process for AAV enrichment by ion-exchange chromatography in our application note AN8008: Real-time monitoring and control of AAV chromatographic enrichment with RT-MALS.
Additional resources
Featured publications:
- Biophysical Characterization of Adeno-Associated Virus Vectors Using Ion-Exchange Chromatography Coupled to Light Scattering Detectors - International Journal of Molecular Sciences (2022).
- Comparison of analytical techniques to quantitate the capsid content of adeno-associated viral vectors - Molecular Therapy: Methods & Clinical Development (2021).
- Comprehensive characterization and quantification of adeno associated vectors by size exclusion chromatography and multi angle light scattering - Scientific Reports (2021).
- Detailed Protocol for the Novel and Scalable Viral Vector Upstream Process for AAV Gene Therapy Manufacturing - Human Gene Therapy (2021).
For additional publications see PL9008: Key publications on the characterization of gene vectors by light scattering
