Can I run IEX- or RPC-MALS experiments on an Eclipse™ equipped with the SEC Switching Option?

Introduction
Eclipse with the SEC Switching Option enables both field-flow fractionation (FFF) and column-based experiments to be run with no manual changes to the fluid connections, making it easy to switch between separation strategies. VISION™, the software for Eclipse control, lets you customize the pump timetable to run chromatographic methods including isocratic size-exclusion chromatography (SEC) as well as gradient techniques like ion-exchange chromatography (IEX) or reverse-phase chromatography (RPC). You can conveniently keep the downstream analytical detectors, such as the DAWN™ and Optilab™, untouched while the Eclipse reconfigures the system between different separation modes.
Advantages of Orthogonal Separation Techniques
While analytical separations by SEC are usually the workhorse methods for macromolecular characterization, FFF is particularly well suited for separating large macromolecules, such as nanoparticles, viruses, and aggregates, that might be filtered off or sheared on a column. It can also be used to reveal true distributions if the sample interacts with the column packing material. Hence FFF complements SEC as an orthogonal method.
Both FFF and SEC fundamentally separate by size. Techniques that separate according to other properties, such as ion-exchange chromatography (IEX) and reverse-phase chromatography (RPC) which separate according to charge or hydrophobicity, are often beneficial for gleaning additional information. These strategies may be particularly advantageous when samples have the same size but have other properties that can be exploited for separation. In this article we will explore how to run these gradient chromatography techniques with the Eclipse and VISION.
First we’ll discuss how to configure a column in VISION, and then how to customize the pump timetable for the solvent gradients needed in IEX- and RPC-MALS.
Step 1: Creating an Eclipse method for SEC experiments
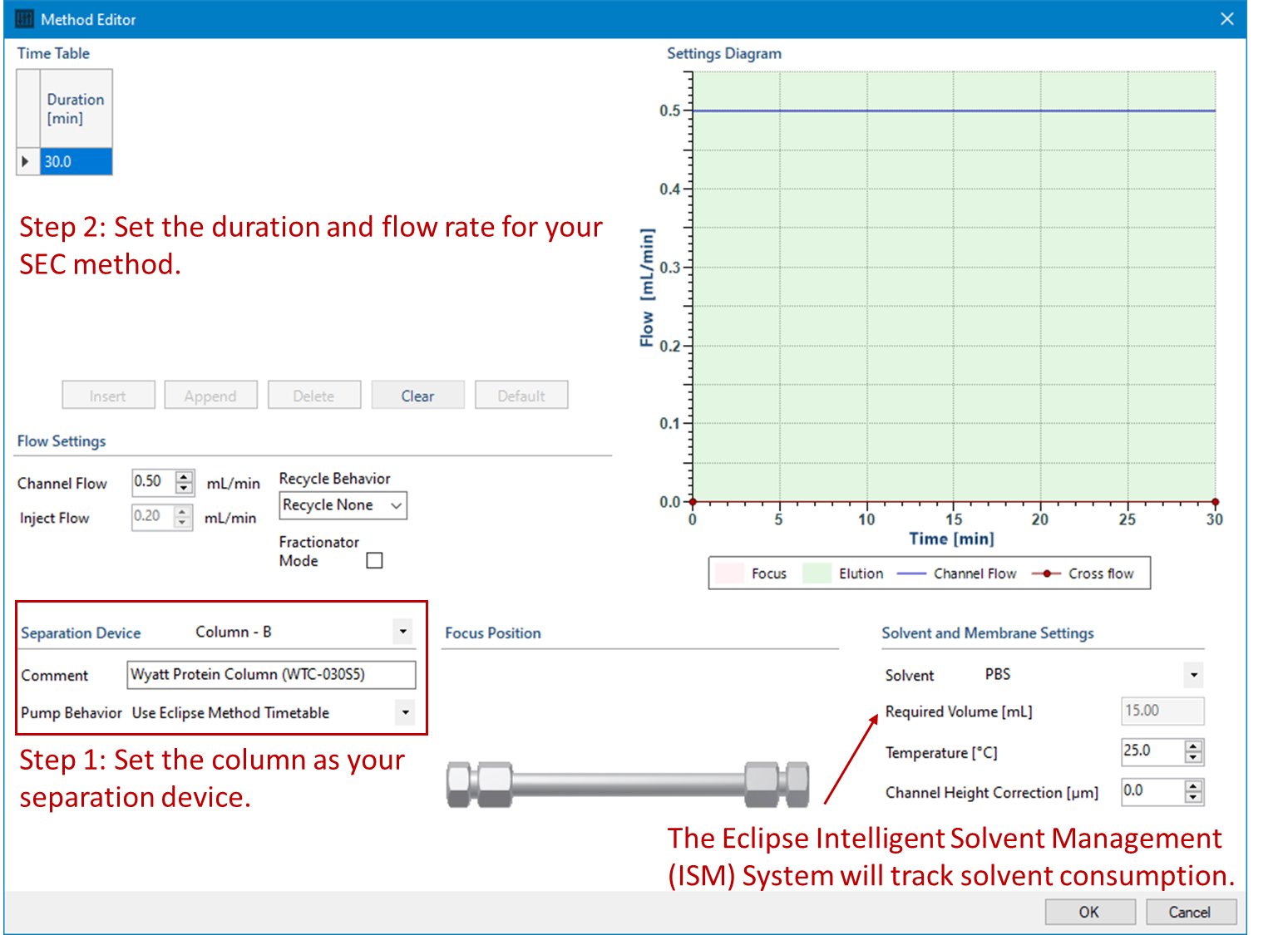
In VISION RUN™, making a method for columns shares a similar workflow to methods with channels that you are already using for FFF experiments. Compared to an FFF method which has a number of different flows and flow rates, SEC mode only requires simple timetable. Select the column as your separation device, then choose the flow rate and duration. The Eclipse Intelligent Solvent Management (ISM) system will track solvent consumption to ensure you won’t run out of solvent in the middle of a sequence.
Additionally, VISION 3 with the Eclipse Switching Option has built-in safeguards to ensure a gradual change in flow rate when switching to (or from) the column in order to mitigate pressure-induced mechanical shock to the columns. This helps minimize ghost peaks from column shedding and improves column lifetime. To improve your MALS data quality even further, consider using columns with low shedding properties, such as the Wyatt Protein or AAV columns.
Step 2: Creating a custom timetable for IEX- or RPC-MALS experiments
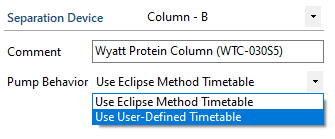
We can customize the pump timetable to produce solvent gradients for IEX- or RPC-MALS, assuming of course that the system employs a binary or quaternary HPLC pump. In order to do so, switch from an Eclipse method to a pump method, which can be done by navigating to the “Pump Behavior” dropdown in the Eclipse method editor and selecting “Use User-Defined Timetable” instead of “Use Eclipse Method Timetable.”
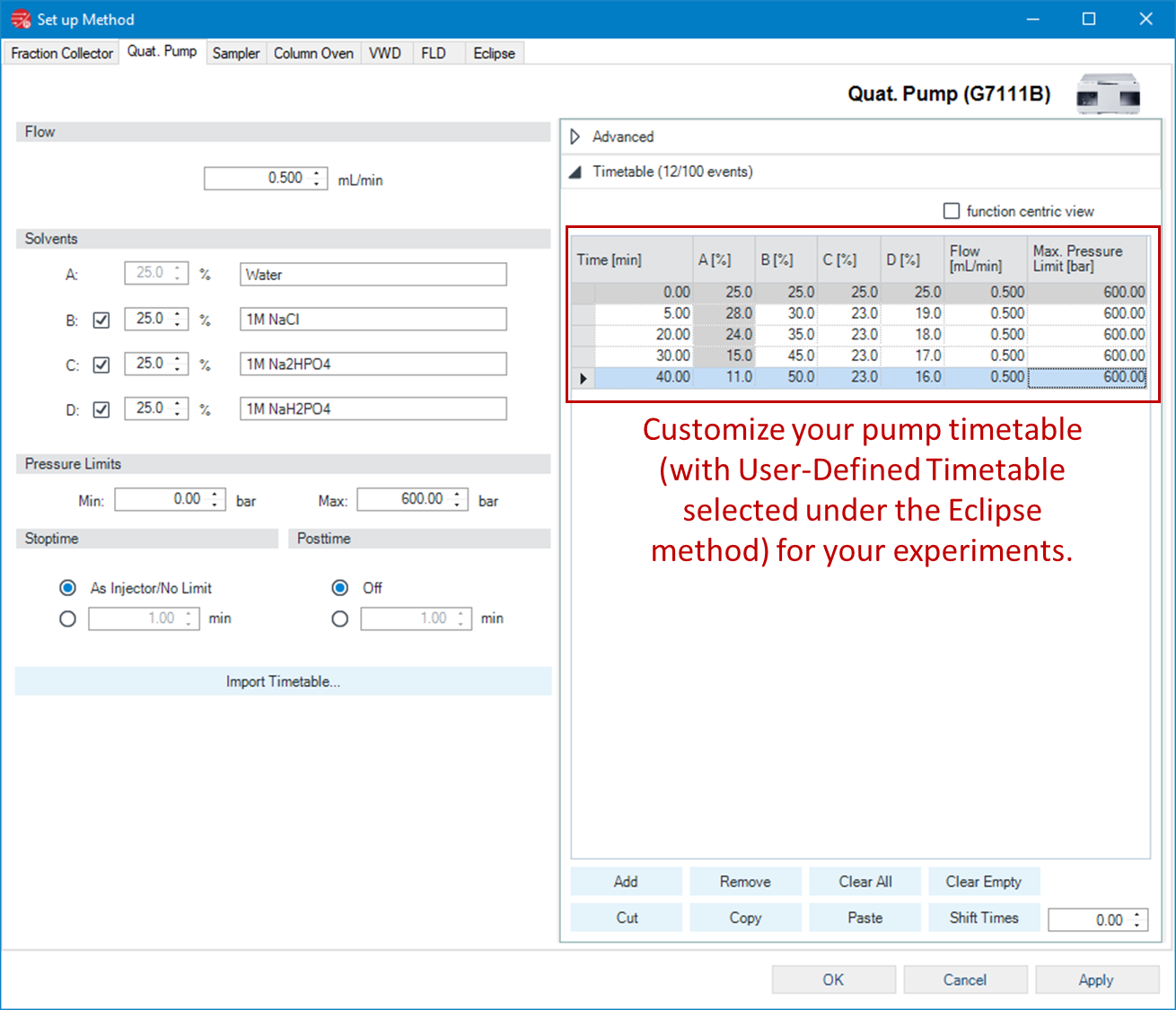
Now that we’ve switched the pump behavior, we can navigate to the pump method and customize the timetable with the desired solvent gradients. Solvent reservoirs can be defined, and rows can be added with various solvent compositions and durations using either the default view or the function-centric view.
Once you’ve built a pump method, as well as any other HPLC module methods, you can save the method, build a sequence, and run all your samples for both FFF and columns in VISION RUN. VISION RUN will generate both VISION DESIGN and ASTRA™ data files for comprehensive analysis.
Some Additional Considerations
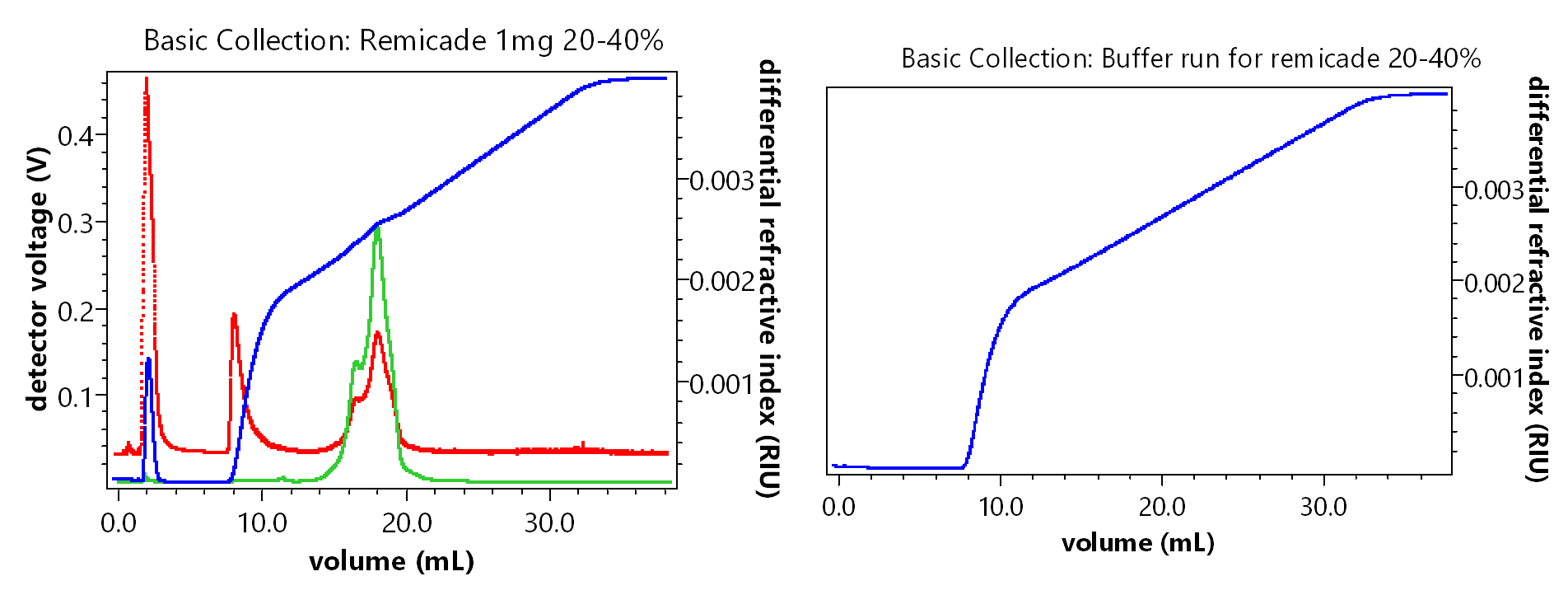
When analyzing experiments with solvent gradients, there are a few things to keep in mind. Any solvent gradient will cause a significant baseline gradient in the differential refractive index (dRI) signal, as you can see in the example ASTRA plots below. A blank injection with the exact same solvent gradient used for the sample should performed, and the data file used to perform baseline subtraction in ASTRA, in order to improve signal-to-noise and account for baseline changes due to the solvent gradient. Baseline subtraction is something you might already be doing for FFF experiments, since cross-flow gradients can lead to drift in the RI signal. These corrections can easily be done in ASTRA with a blank run.
Conclusion
If you regularly work with large solvent gradients, the Optilab High Concentration model is the only dRI instrument that can handle many of the gradients used in IEX-MALS. For example, the standard Optilab can measure gradients between 0 – 450 mM NaCl without saturating, whereas the high concentration model can measure gradients up to 3.4 M NaCl.
Do you have a question? Contact our experts here in Customer Support. We’re happy to help! Call +1 (805) 681-9009 option 4.