Understanding Flow Field-Flow Fractionation
Asymmetric-Flow Field-Flow Fractionation (AF4)
Asymmetric flow field-flow fractionation is a versatile separation technique that can be applied to macromolecules and nanoparticles from 1 – 1000 nm and beyond, implemented in Eclipse™.
How AF4 separates by size
Structure of the AF4 channel
The AF4 principle, described in Figure 1, was described by Giddings in a review article1. Briefly, separation takes place in an open channel consisting of two long, narrow blocks bolted together with a spacer in between. The spacer is a polymer foil with a typical thickness of just 100 to 500 µm. Flow within this thin ribbon-like channel is laminar, with a pronounced parabolic flow profile that drives particle separations.
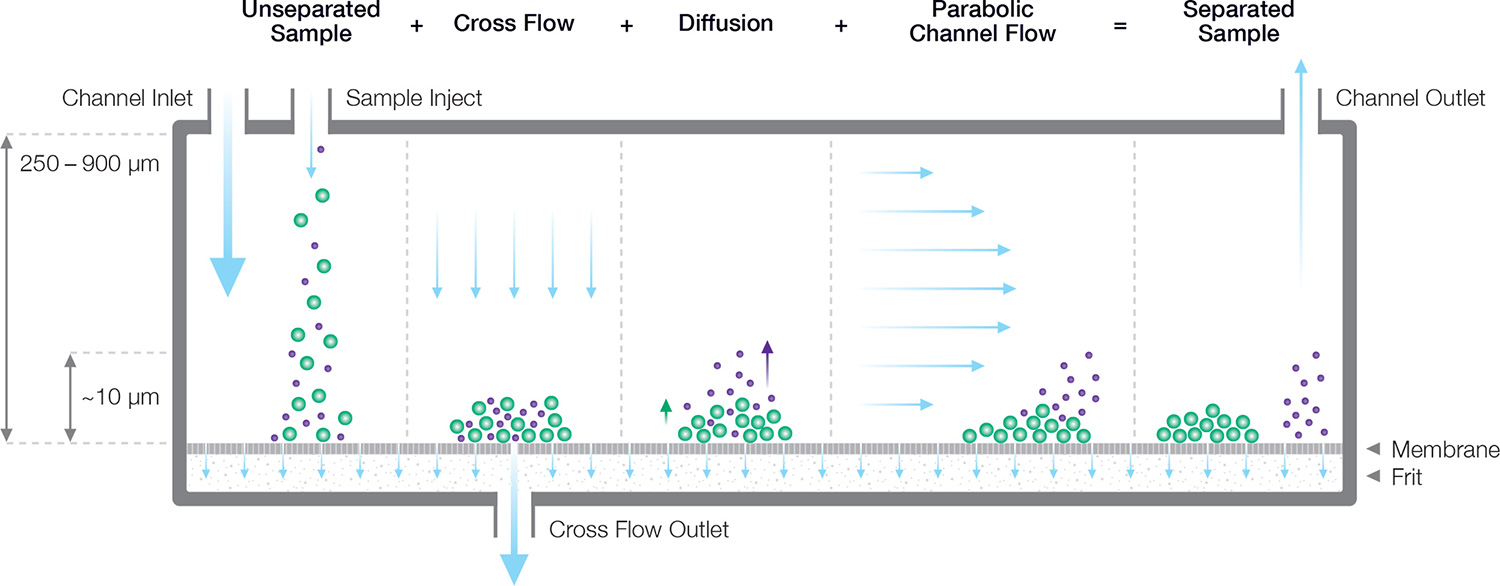
Figure 1. Separation mechanism in an FFF channel, consisting of a semi-permeable membrane and a controllable lateral flow restrictor which forces some of the solvent to flow through the membrane. The size-dependent balance between diffusion and cross flow leads to different height distributions for different sizes. Smaller particles are swept out of the channel earlier than larger particles, which remain close to the membrane and experience a lower flow velocity.
The bottom block comprises a semi-permeable membrane supported by a frit. The membrane is permeable to solvent but not to the analyte; this essential function is guaranteed by selecting the appropriate membrane pore size, expressed as a molecular-weight cutoff (MWCO) ranging from 1 kDa to 100 kDa.
AF4 principle
The particle-containing carrier fluid flows parallel to the membrane, but a constriction at the end of the channel forces some of the fluid to pass through the membrane, creating a “cross-flow field” that concentrates the particles towards the membrane. Diffusion acts as a counterforce that drives particles back up into the channel, resulting in a height distribution above the membrane that depends on the particle’s translational diffusion coefficient Dt, and hence hydrodynamic radius Rh, as well as the cross-flow velocity2. Separation arises from differential transport velocity in the laminar flow profile according to the particles’ height above the membrane.
In FFF, the particle retention time tR depends on Dt, the channel thickness w, the cross-flow rate Fc (which is controlled by a precise flow controller), and the detector flow rate Fout. If the flow rates are constant over time and retention is sufficiently high, the retention time is, in good approximation, given by Eq. 12:
tR = w2/6Dt x ln(1+ Fc/Fout) Eq. 1
Retention time, zone broadening and dilution of the sample at the channel outlet for more complex flow profiles can be calculated by standard fluid dynamics3. Computer simulation of the separation process enables virtual optimization of the separation method, as well as calculation of diffusion coefficients based on the measured retention time4. An example of the excellent separation capabilities of FFF is shown in Figure 2.
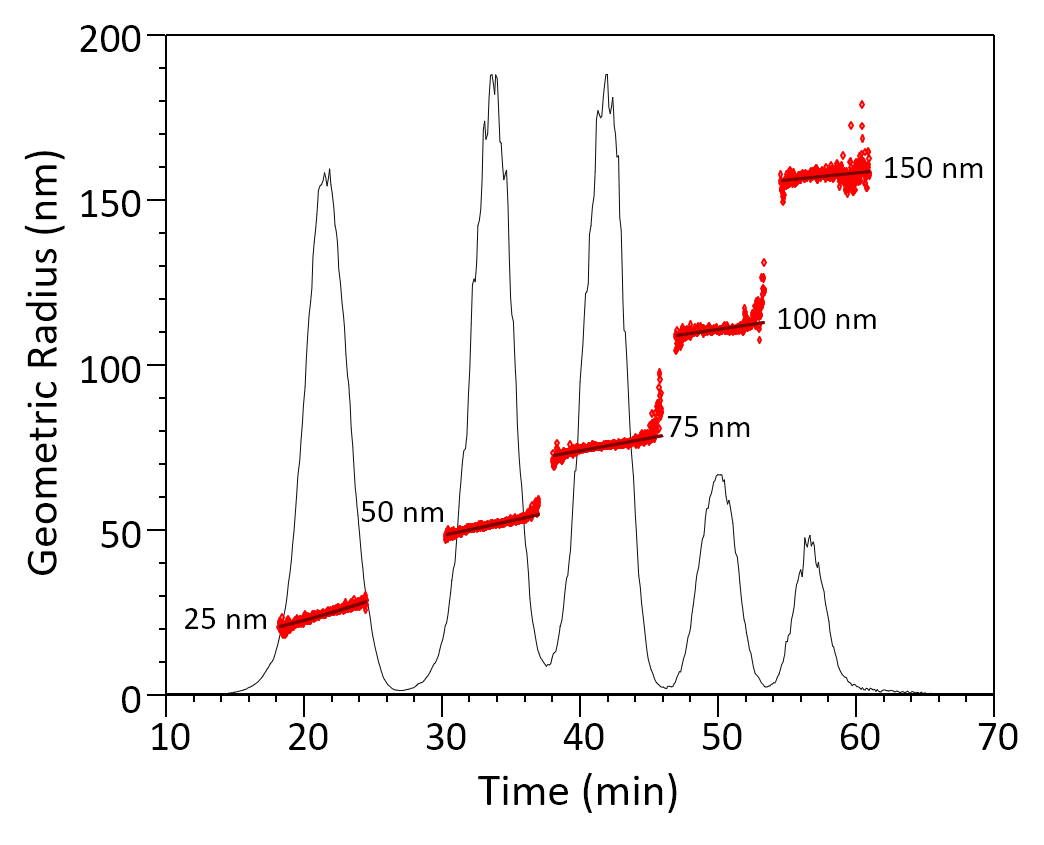
FFF separation of a mix or particles, illustrating excellent resolution. The average size and heterogeneity of each population is calculated from size of each eluting fraction (slice).
AF4 procedure
With AF4 separation there is no column media to interact with the samples, so for very high molar mass polymers, there is no need to worry about shearing forces being applied. The entire separation is gentle, rapid, and non-destructive without a stationary phase that may interact, degrade, or alter the sample.
The separation process requires three steps: During the first two steps, injection and focusing, the main flow is split, enters the channel from both ends. At this point the flow will move only down and through the membrane. When the sample is injected it is focused in a thin band and concentrated towards the membrane. The position of that band depends on the ratio of flow rates from the inlet and outlet ports – higher flow from the inlet port moves the focus position downstream.
Two modes of injection are available: ‘tip injection’ and ‘focus-zone injection’.
- Tip injection: the sample is introduced via the inlet port and flows along the membrane until reaching the focus position. This has the advantage of a simpler channel design but the disadvantage of possible loss of sample during focusing. Eclipse channels do not use this mode, but it is available in the Eclipse controller for use with SEC columns.
- Focus-zone injection: the sample is injected independently of the inlet flow, via a separate port located directly above the focusing position. The ratio of flows from the inlet port and outlet port is balanced to meet directly under the injection port. This requires an extra injection port but has the benefit of fully retaining the sample during focusing. All Eclipse channels except the Dispersion Inlet Channel (which does not focus the sample) operate in this mode.
After complete transfer of the sample volume the injection flow is stopped and one typically allows for another minute of focusing before the flow pattern is switched to the elution mode.
In the elution step, the flow enters only from the inlet port and exits at the outlet which is connected to the detectors. Sample constituents elute separated according to size and are monitored by the array of detectors.
1 Giddings, J. C. A New Separation Concept Based on a Coupling of Concentration and Flow Nonuniformities. Sep. Sci. 1, 123–125 (1966).
2 Wahlund, K. G. & Giddings, J. C. Properties of an Asymmetrical Flow Field-Flow Fractionation Channel Having One Permeable Wall. Anal. Chem. 59, 1332–1339 (1987).
3 Litzen, A. & Wahlund, K. G. Zone broadening and dilution in rectangular and trapezoidal asymmetrical flow field-flow fractionation channels. Anal. Chem. 63, 1001–1007 (1991).
4 Elsenberg, S. & Johann, C. Field-Flow Fractionation: Virtual Optimization for Versatile Separation Methods | LCGC. The Column http://www.chromatographyonline.com/field-flow-fractionation-virtual-optimization-versatile-separation-methods-0 (2017).
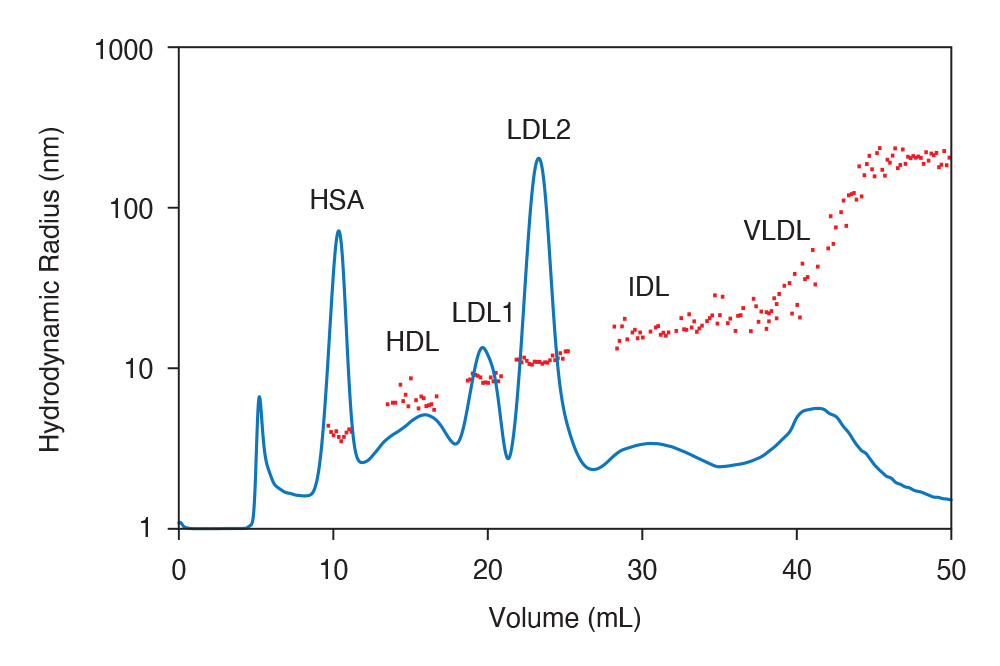
AF4-MALS-DLS separation of whole serum, with distinct peaks for serum albumin, IgG, and various types of lipoproteins. Hydrodynamic radii (Rh) were determined by online dynamic light scattering embedded in the DAWN MALS detector.
Electrical/Asymmetric-Flow FFF (EAF4)
EAF4 incorporates an additional electrical field, applied perpendicular to the membrane. Charged particles exhibit a shift in height above the membrane, and a corresponding shift in retention time, that varies with applied field strength, from which the electrophoretic mobility µE and zeta potential may be calculated. This method can reliably determine µE for several components simultaneously, providing an indication of the charge distribution5. EAF4 is implemented in Eclipse equipped with a Mobility™ module and channel.
5 Johann, C., Elsenberg, S., Schuch, H. & Rösch, U. Instrument and Method to Determine the Electrophoretic Mobility of Nanoparticles and Proteins by Combining Electrical and Flow Field-Flow Fractionation. Anal. Chem. 87, 4292–4298 (2015).
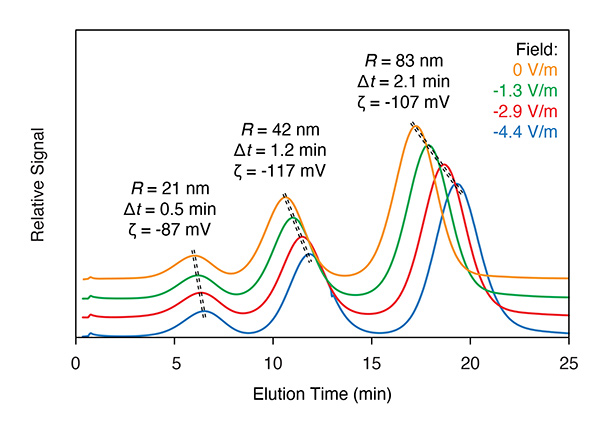
In EAF4, the shift in retention time due to the electrical field is analyzed to determine zeta potential and electrophoretic mobility of each eluting peak.
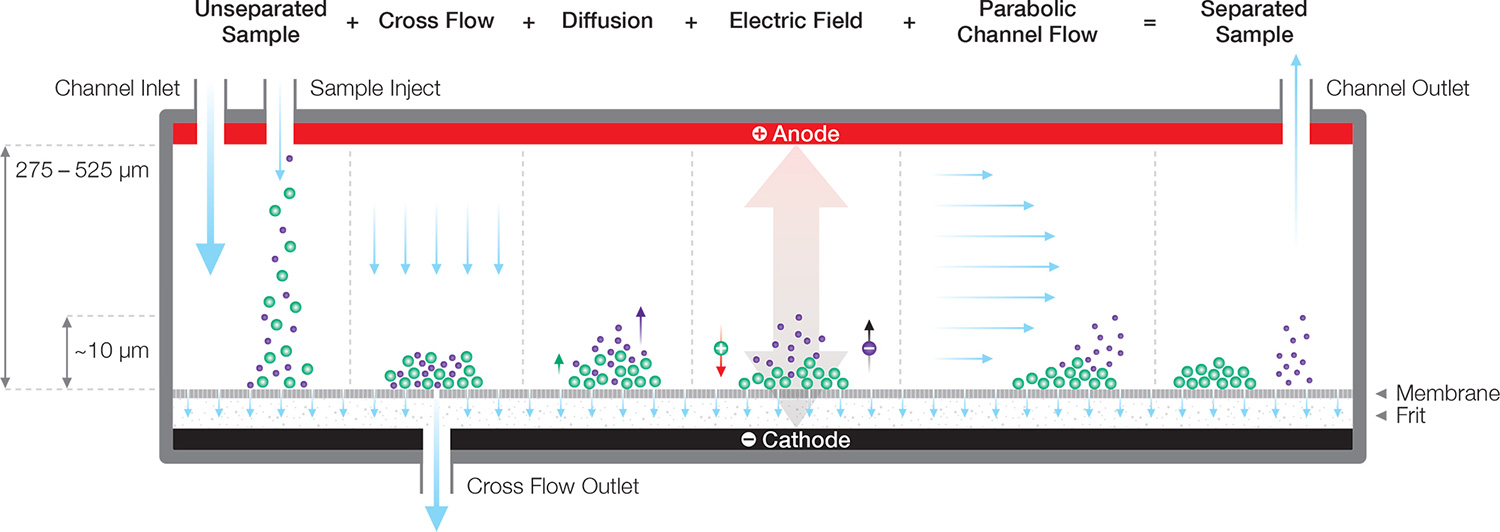
Separation in EAF4 occurs according to both size and charge. For charged particles, the equilibrium height above the membrane (and therefore retention time) is shifted according to the sign and magnitude of the applied field.
