Particle Concentration
Introduction
Light scattering measures the concentration of large ensembles of nanoparticles, overcoming the limitations of single-particle counting methods such as electron microscopy, NTA or TRPS that provide low statistical sampling yet require stringent sample preparation and dilution to a narrow concentration window.
Furthermore, light scattering instruments are not clogged by larger-than-expected particles, and the only manual labor involved is usually just pipetting or dispensing into a vial, cuvette or microwell plate.
The light scattering techniques Wyatt provides for concentration analysis are based on multi-angle light scattering (MALS) and dynamic light scattering (DLS). They trade off resolution, speed and sample volume, in order to meet the requirements of high-throughput screening, detailed characterization or real-time operation.

SEC-MALS and FFF-MALS provide quantitative and accurate particle concentrations and size distributions. Measurements are fully automated with industry-standard autosamplers and pumps.
How light scattering measures PC
Particle concentration (PC) is calculated from scattered intensity, particle volume and the refractive indices nparticle, nsolvent of the particle material and the solvent.

- In addition to scattered intensity, both MALS and DLS determine particle size, from which the particle’s volume is calculated.
- Refractive indices are taken from the literature or calculated from the particle composition if it is not homogeneous; the particle’s effective nparticle may also be calibrated against a known concentration of identical particles.
That’s all it takes! More details may be found in the MALS theory and DLS theory pages.
Applications
The size range, convenience and automation inherent in MALS and DLS make them well-suited to determining concentration and characterizing quantitative particle size distributions of nanomedicines and engineered nanoparticles such as:
- viruses and viral vectors
- lipid nanoparticles and liposomes
- polymersomes, PLGA nanoparticles
- nanobeads for bioprocessing and nanocages
- decorated gold nanoparticles
These techniques may be applied in research and discovery, product and process development, production and quality control, in standard labs or GMP environments.

The DynaPro Plate Reader measures particle size and concentration in standard flat-bottom 96, 384 or 1536 microwell plates.
Methods
SEC/FFF-MALS
MALS is most often coupled downstream of a size-based separation method such as size-exclusion chromatography (SEC-MALS) or field-flow fractionation (FFF-MALS). Separation prior to measurement enables high-resolution particle size distributions across a size range of 13 nm to 650 nm in radius (equivalent to rms radius range of 10 nm to 500 nm), where the resolution is limited mainly by the separation method.
Because each size fraction is measured individually, the detector is not “blinded” to small particles by strong scattering from large particles so concentration values are generally quite accurate. If particles composed of different materials are separated, each peak may be assigned a corresponding nparticle value. SEC-MALS uses a standard HPLC system with a DAWN™ MALS instrument; FFF-MALS adds an Eclipse™ FFF controller and separation channel. ASTRA™ software analyzes the particle size and concentration in each eluting fraction, and can integrate peaks to calculate the total number of injected particles in each size range. ASTRA supports 21 CFR Part 11 compliance, as does the VISION™ FFF software.
In addition to size and concentration, a setup may be configured with UV, fluorescence and/or refractive index detectors in order to determine the molar mass, composition and payload of many types of nanoparticles.
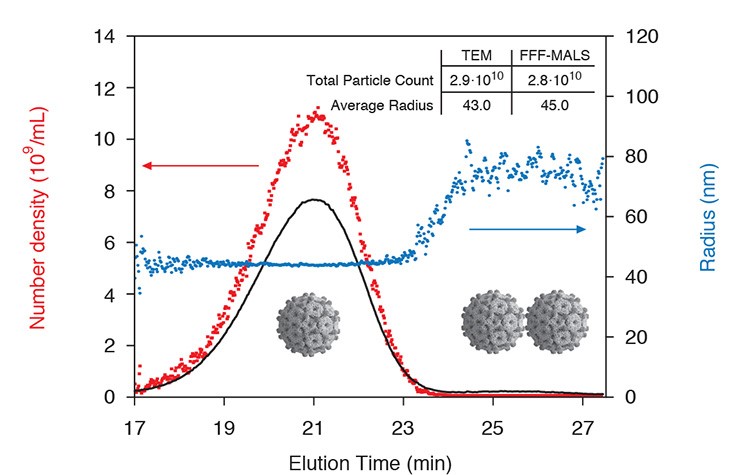
FFF-MALS is an excellent tool for vaccine characterization. Total viral count and size may be quantified with high resolution for each subpopulation.
MALS for PAT
Real-time MALS (RT-MALS) measurements of concentration are generally performed without separation in order to provide rapid feedback for process analytical (PAT) applications, reporting results 30 times per minute. The values may be integrated to calculate the final titer of, e.g., virions in the pooled fractions. At the same time, size is determined continuously for use as a biologics DSP pooling criterion or to monitor deviations of synthetic nanoparticles in the course of manufacturing.
The ultraDAWN™ RT-MALS instrument is usually connected in-line with production or purification systems operating at lower flow rates. For static reactors or high-flow-rate processes, it is configured on-line, where a pump draws some of the product and runs it through the ultraDAWN. OBSERVER™ software orchestrates the entire process; pre-defined trigger conditions may be set to alert the operator upon reaching an end point or deviating from acceptable product attributes.
RT-MALS only determines the average size and not a full distribution, so if the size distribution is wide, the particle concentration values may not be accurate. The reported concentration is most reliable for narrowly disperse nanoparticles.
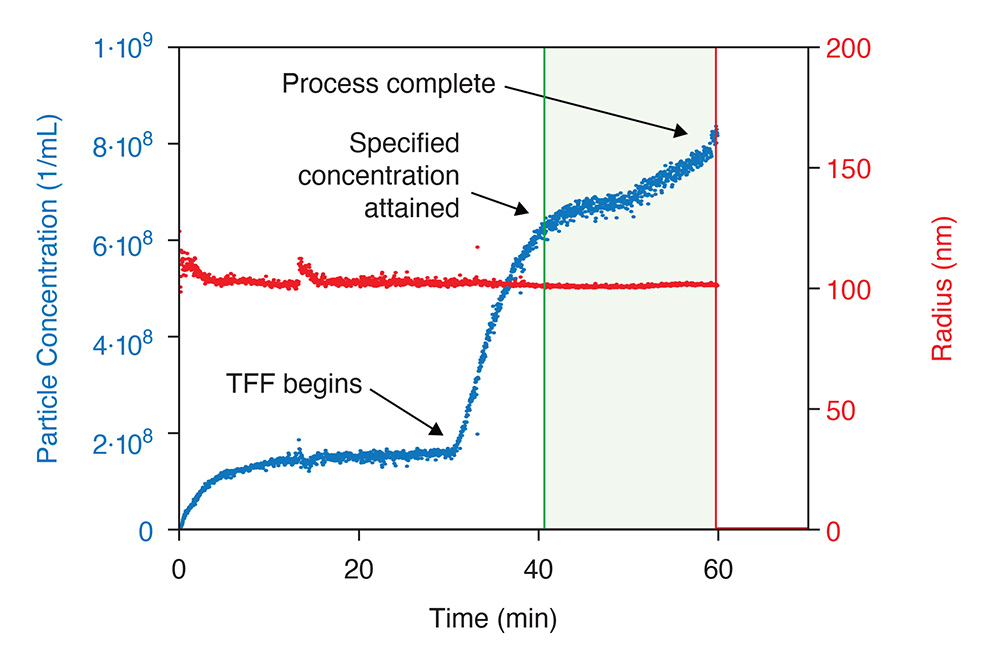
RT-MALS monitors particle concentration in the course of tangential flow filtration. A trigger upon reaching the specified end point. The ultraDAWN simultaneously measures particle size to assess virion degradation.
DLS
DLS is usually performed without separation, in microcuvettes or microwell plates. This “batch” mode operation enables fast measurements of relatively low-resolution distributions, with less accuracy than SEC-MALS or FFF-MALS but potentially more than RT-MALS. High-throughput DLS in microwell plates is ideal for analyzing viral vector downstream bioprocess fractions or a nanoparticle formulation matrix.
The size range (hydrodynamic radius) over which concentration may be measured is 0.5 nm to 160 nm in the DynaPro™ Plate Reader and 0.2 nm to 175 nm in the DynaPro™ NanoStar™, making it suitable for most viruses and nanomedicines as well as other types of nanoparticles. If more than one size population is evident in the distribution, DYNAMICS™ software will provide the concentration of each.
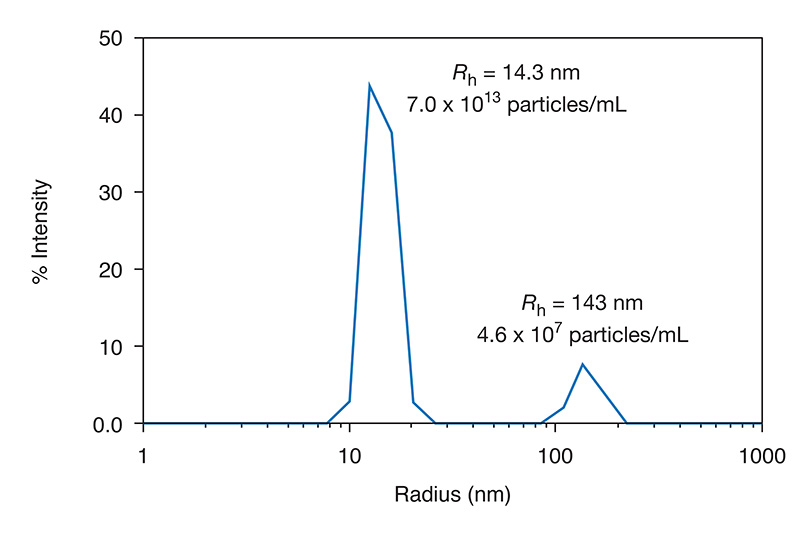
Size distribution obtained by DLS showing the mean size of two populations and the concentration of each.
Wyatt’s DLS instruments dramatically minimize sample requirements. Only 2 µL of sample are required in quartz cuvettes, and in the most common 384 well plates, 20 µL are needed for optimal measurements. Though these instruments cover several orders of magnitude in concentration, if particle concentrations are high and dilution is necessary, it is easy to do so directly in the measurement volume.
DLS in QC and at-line PAT
DLS is especially useful for quick and easy nanoparticle analysis with minimal instrument complexity or sample prep effort. Wyatt’s DLS instruments are supported by DYNAMICS™ software, a comprehensive suite of instrument control, data acquisition and analysis that provides extensive automation, rich characterization capabilities and intuitive feedback on the quality and reliability of each and every measurement.
For use in quality control and at-line process analytics, DYNAMICS offer 21 CFR Part 11 compliance. In addition, it supports an application programming interface (API) that enables integration with third-party liquid handlers and high-level process or quality control software.

Resources
Selected References
Improved multidetector-asymmetrical flow field flow fractionation method for particle sizing and concentration measurements of lipid-based nanocarriers for RNA delivery, Mildner, R. et al. Eu. J. Pharm. Biopharm. 163, 252-265 (2021) https://doi.org/10.1016/j.ejpb.2021.03.004
Biophysical characterization of influenza virus subpopulations using field flow fractionation and multiangle light scattering: correlation of particle counts, size distribution and infectivity, Wei, Z. et al J. Virolog. Meth. 144(1-2), 122-132 (2007). https://doi.org/10.1016/j.jviromet.2007.04.008
Quantitation of influenza virus using field-flow fractionation and multi-angle light scattering for quantifying influenza A particles, Bousse, T. et al. J. Virolog. Meth. 193(2), 589-596 (2013). https://doi.org/10.1016/j.jviromet.2013.07.026
Quantification and characterization of virus-like particles by size-exclusion chromatography and nanoparticle tracking analysis, Steppert, P. et al. J. Chromat. A 1487, 89-99 (2017) http://dx.doi.org/10.1016/j.chroma.2016.12.085