Biotherapeutics
Multi-Angle Light Scattering (MALS) and high-throughput Dynamic Light Scattering (DLS) instruments provide powerful tools for characterizing biomolecule candidates across many excipients and buffer conditions.
Introduction

Development of efficacious and robust biologics is a complex process. Light scattering technologies assist at each stage with uniquely versatile technologies for biophysical screening and characterization, from target and candidate discovery to selection, optimization, purification, formulation and manufacturing scale-up.
Some key applications of light scattering in biotherapeutic development are described below.
Target & Candidate Proteins
Once target proteins and candidate biomolecules for a given indication have been identified, they must be produced at a small scale in sufficient quantities and appropriate quality for further R&D.
Soluble aggregates
Size exclusion chromatography with multi-angle light scattering (SEC-MALS) accurately characterizes proteins and other biomolecules for soluble aggregates, regardless of non-ideal column interactions, to quickly identify optimal purification conditions.
Stability
High-throughput dynamic light scattering (DLS) using the DynaPro™ Plate Reader requires minimal sample quantities and time to assess candidates and pre-formulation conditions for stability and propensity for aggregation.
Interactions
Composition-gradient, multi-angle light scattering (CG-MALS) combining a Calypso™ composition-gradient system and DAWN™ MALS detector determines affinity and absolute stoichiometry of drug-target binding, without labeling or immobilization. It is particularly well suited for studying multivalent interactions or drug-target binding in the presence of self-association, including virus-like particles (VLPs), monomeric antibodies, oligonucleotides or peptides.
Candidate Selection & Optimization
Post-translational modifications such as glycosylation or PEGylation are often applied to proteins or peptides in order to improve stability and increase physiological retention time. Some biologics, such as insulin, are required to form well-defined oligomeric states for the same reason. Modification of IgG with conjugated, highly toxic drugs can convert an antibody to a potent anti-cancer substance.
Drug-Antibody Ratio
Triple-detection SEC-MALS utilizing combined UV, MALS and RI instruments readily determines the degree of glycosylation or PEGylation, and in many instances may be used to calculate the drug-antibody ratio (DAR) of antibody drug conjugates (ADCs).
Oligomerization
CG-MALS analyzes reversible oligomerization by measuring the dependence of weight-average molar mass on concentration. The oligomeric order may be determined unambiguously, along with the affinity.
Antibody engineering and other modifications by means of mutations to the genetic sequence may be carried out in order to improve stability or efficacy. SEC-MALS helps determine the impact of these changes on aggregation, while CG-MALS assesses how they have affected target binding properties.
Biocolloids
For larger entities such as virus-like particles (VLPs) and polymer, liposome or hydrogel-based drug carriers, separation by field-flow fractionation (FFF) is usually preferable to size exclusion chromatography. The Eclipse™ FFF system offer a variety of options for optimal separation, upstream of a MALS detector.
Process Development
Light scattering measurements help process development in several ways:
Purification end point
Since multi-angle light scattering determines molar masses independently of retention time, RT-MALS is ideal for real-time characterization of purification products and determination of end points. For more detailed product characterization during process development, SEC-MALS may be implemented at-line with UHPLC for rapid separation and analysis.
Quaternary structure
Complex products such as virus-like particles or peptide micelles must be produced with specific masses, shapes and sizes, as well as minimum quantities of residual unassembled material. SEC-MALS or FFF-MALS with on-line DLS validates such processes to quantify unassembled sub-units and ensure that the final product conforms to the required structure.
Formulation
In the formulation process, many combinations of buffer conditions and excipients must be tested for stability and propensity for aggregation. Final selection of the most promising conditions for long-term testing does not necessarily require rigorous thermodynamic characterization, but does benefit from an understanding of multiple properties which reflect various facets of stability. High-throughput DLS is the ideal technique for this, as it can address multiple stability-indicating parameters simultaneously testing thousands of conditions per day:
Stability screening
- Aggregation state - formation of aggregates, size distribution and rate of formation. This is the basic DLS analysis.
- Thermal stability - how temperature induces denaturing and non-native aggregation. Determined through temperature ramps, monitoring changes in molecular size.
- Chemical stability - how buffer composition (pH, ionic strength, denaturing solvents such as guanidine) induces denaturing and non-native aggregation. Determined through composition steps, monitoring changes in molecular size.
- Colloidal stability - how surface moieties such as charged residues or hydrophobic patches lead to aggregation, unrelated to changes in tertiary structure. Determined through the concentration dependence of translational diffusion, kD or D1.
With the DynaPro Plate Reader, these measurements takes place in situ in standard 96, 384 or 1536 microwell plates, consuming minimal sample quantities. The same samples may be further tested in the same or other plate-based instruments [e.g., to analyze the temperature dependence of colloidal stability (in the DynaPro) or intrinsic fluorescence (in a spectroscopic plate reader)], making the DynaPro Plate Reader an invaluable tool for formulation screening. A software package enables integration of the instrument with robotic sample preparation and plate placement.
Molecular charge
Another helpful light scattering tool for formulation development is Electrophoretic Light Scattering (ELS). The Mobius™ integrates with an autosampler and the Atlas™ for automated measurement of molecular charge, an important parameter for stability. The measurement takes place in native buffers, even under moderate to high salt conditions. Like the DynaPro Plate Reader, the Mobius offers automated measurement of multiple parameters such as thermal and colloidal stability.
Turbidity/opalescence
Opalescence indicates aggregation and particle formation, reversible self-association (RSA), and liquid-liquid phase separation that leads to undesirable product properties. The DynaPro™ NanoStar™ with microliter volumes provides quick turbidity/opalescence measurements of formulations.
High-Concentration Formulation
Additional characterization challenges in the course of high-concentration formulation of monoclonal antibodies and other biotherapeutics include understanding the interplay of weak interactions, and viscosity.
Protein-Protein interactions
Weak protein-protein interactions may not be important at low concentrations, and so are not necessarily reflected in dilute-condition parameters such as kD, but come to dominate at high concentration. CG-MALS uniquely addresses these complex interactions through a theoretical framework known as the Minton-Chatelier formalism. This technique distinguishes attractive interactions causing aggregation, opalescence and viscosity, from repulsive interactions which may impact measurements but have little effect on stability.
Viscosity screening
Viscosity is both indicative of colloidal stability, and an important parameter on its own since high viscosity impacts manufacturability and deliverability of the final product. DLS readily determines viscosity of concentrated protein solutions using tracer beads. The DynaPro Plate Reader can evaluate the viscosity of samples as small as 10 µL in a few seconds, measuring thousands of conditions per day in order to screen for ideal viscosity-reducing excipients.
Turbidity or opalescence measurements
Turbidity becomes important in high-concentration formulations due to interactions between different species in solution resulting in opalescence and poor product quality. Turbidity of a solution is interpolated from a turbidity calibration plot. A quartz cuvette is calibrated against a series of turbidity standards, and the turbidity of a solution subsequently measured in that cuvette is calculated and displayed in the data table in nephelometric units (NTU). Visit our turbidity page to learn more.
Biophysical Characterization
Light scattering is an essential part of any biophysical characterization lab for biotherapeutics, and is recognized by the FDA as a key technology for regulatory filings. Key applications include:
Aggregation
Applying orthogonal techniques based on light scattering and other instruments provided by Wyatt, analytical support labs can support regulatory filings with thoroughly validated, extended analysis of soluble and insoluble sub-micron aggregates. These techniques are:
- SEC-MALS for robust characterization of the mass and size distributions of soluble, irreversible aggregates
- FFF-MALS for robust characterization the mass and size distributions of soluble and insoluble, irreversible aggregates
- Batch DLS for rapid testing of size distributions for soluble and insoluble, reversible and irreversible aggregates
- CG-MALS for characterization of reversible aggregates or oligomers, as well as validation of SEC-MALS for loss of aggregates on the column.
Conjugation
Triple detection by UV-MALS-dRI determines the composition as well as molar mass distributions of:
- Protein-polysaccharide conjugated vaccines
- PEGylated proteins
- Antibody-drug conjugates
- glycoproteins
- VLPs with DNA/RNA payloads
With the addition of a WyattQELS™ DLS module, the size distributions may be determined as well.
- Chemical disposition: ELS determines charge state and so is an important indicator of surface chemistry.
- Functionality: CG-MALS is a powerful technique for in-depth characterization of biomolecular interactions such as drug-target binding, without labeling or immobilization. Since it measures molar mass directly, it is particularly versatile for a range of valencies and self- as well as hetero-association.
Vaccines
The universe of vaccines is highly diverse, covering traditional vaccines based on live-attenuated or inactivated viruses and through the most advanced modalities utilizing gene therapy. Wyatt’s suite of solutions is essential in all stages of vaccine development, from basic virology through product characterization, production and quality control. Visit Solutions for vaccine characterization and development to learn more.

Process Analytical Technology
In current biotherapeutic manufacturing strategies, “the process is the product” means that strict control of process parameters such as pH, temperature, and feedstock consumption is required to guarantee product quality. Only at the end of the process can QC procedures check the critical quality attributes (CQAs) that define product quality. Hence, most PAT instruments monitor process conditions rather than product attributes.
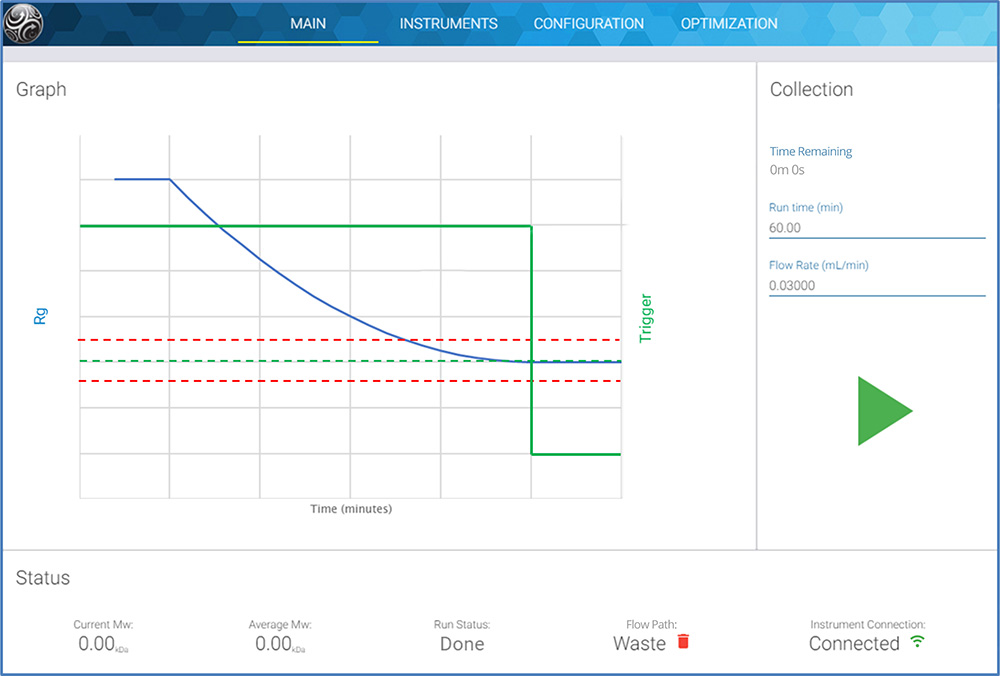
Real-time multi-angle light scattering (RT-MALS) is a novel PAT application of light scattering, that directly determines the molecular weight of mAbs and other therapeutic biomolecules. It is particularly sensitive to the appearance of aggregates and contaminant particles. Using the ultraDAWN™ RT-MALS instrument, in-line or on-line with purification and other process steps, production staff can speed up and optimize process development with immediate feedback on the impact of process variations and deviations. UltraDAWN measures another attribute, the average radius, for virus-like particles and other biomolecular complexes with diameters above 25-30 nm.
After developing the process, the ultraDAWN may be left on-line. Its accompanying software, OBSERVER™, integrates with an OPC-UA environment for automated and/or GMP operation. OBSERVER can be programmed to trigger the system to react to deviations from the allowed molar mass range due to aggregation, or to shut down a reaction once the endpoint has been reached.
Resources
Application notes
Selected References
Esfandiary, R.; Hayes, D. B.; Parupudi, A.; Casas-Finet, J.; Bai, S.; Samra, H. S.; Shah, A. U.; Sathish, H. A. A systematic multitechnique approach for detection and characterization of reversible self-association during formulation development of therapeutic antibodies. J. Pharm. Sci. 2013, 102, 3089-3099.
Lehermayr, C.; Mahler, H.-C.; Mäder, K.; Fischer, S. Assessment of net charge and protein-protein interactions of different monoclonal antibodies. J. Pharm. Sci. 2011, 100, 2551-2562.
Menzen, T.; Friess, W. Temperature-ramped studies on the aggregation, unfolding, and interaction of a therapeutic monoclonal antibody. J. Pharm. Sci. 2014, 103, 445-455.
Mohr, J.; Chuan, Y. P.; Wu, Y.; Lua, L. H. L.; Middelberg, A. P. J. Virus-like particle formulation optimization by miniaturized high-throughput screening. Methods 2013, 60, 248-256.
Rathore, A.; Krull, I. S.; Kreimer, S. The Use of Light-Scattering Detection with SEC and HPLC for Protein and Antibody Studies, Part I: Background, Theory, and Potential Uses. LCGC North America 2012, 30(9).
Rathore, A.; Krull, I. S.; Kreimer, S., Champagne, J. The Use of Light-Scattering Detection with SEC and HPLC for Protein and Antibody Studies, Part II: Examples and Comparison to Mass Spectrometry. LCGC North America 2012, 30(12).
Saito, S.; Hasegawa, J.; Kobayashi, N.; Tomitsuka, T.; Uchiyama, S.; Fukui, K. Effects of ionic strength and sugars on the aggregation propensity of monoclonal antibodies: influence of colloidal and conformational stabilities. Pharm. Res. 2013, 30, 1263-1280.
