Solutions for Nanoparticle Characterization
Wyatt’s Light Scattering Toolkit provides essential analytical instrumentation for nanoparticle characterization.
Introduction
Nanoparticles are diverse and ever-growing in importance. Their characterization covers a wide range of challenges including:
- Determination of size, concentration, content and conformation of nanomedicines including drug and gene delivery vehicles
- Stability assessments of colloidal suspensions or nanoemulsions for controlled-release applications
- Detection and identification of small quantities of industrial additives that may leach into the environment
- Analysis of size distributions, zeta potential, and aggregation behavior dependent on solution conditions, including pH and ionic strength, of nanoparticles, emulsions and colloids utilized in food, cosmetics, semiconductor processing and a host of other consumer or industrial products
Wyatt’s Light Scattering Toolkit provides essential analytical instrumentation for nanoparticles combined with unparalleled levels of automation, sensitivity and resolution. By turning commonly utilized, low-throughput manual measurements into automated, high-throughput workflows, a plethora of new possibilities are opened to scientists and engineers working in this field.
The measurement techniques applied to nanoparticle and colloid characterization in Wyatt instruments are dynamic light scattering (DLS), multi-angle static light scattering (MALS), field-flow fractionation (FFF) and electrophoretic light scattering (ELS).
Properties
Size Distributions
DLS is a common means for estimating the size of nanoparticle and other colloidal suspensions, and even obtaining a low-resolution size distribution, in all kinds of solvents and in relatively short time.
However, the majority of DLS instruments require you to manually fill a cuvette with sample, insert the cuvette into the instrument, operate the software to begin data collection, then wash the cuvette and repeat the entire procedure with the next sample – a slow and laborious process. Wyatt offers two methods of automating DLS so you can set and forget dozens or even hundreds of samples, freeing you up to do other things and increasing productivity ten-fold.
- The DynaPro™ Plate Reader performs DLS measurements in situ in standard microwell plates containing 96, 384 and even 1536 samples, completing hundreds or thousands of samples per day.
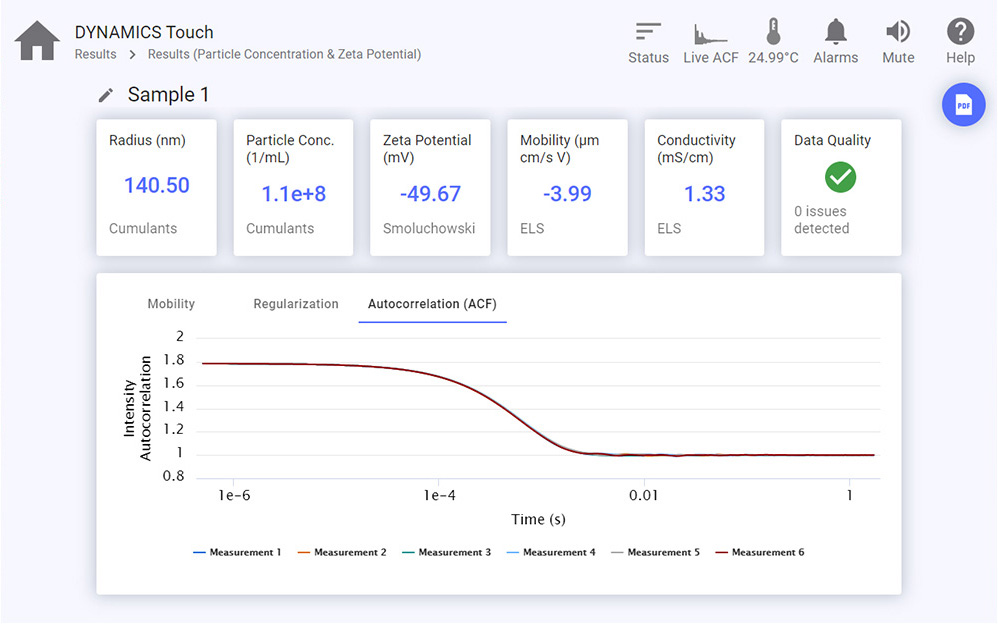
- The ZetaStar™, combining simultaneous DLS and electrophoretic mobility measurements to determine size and zeta potential, can be connected to standard autosamplers and HPLC pumps in order to automate dozens of size and zeta potential measurements. The ZetaStar works with organic and aqueous solvents.
- If automation is not needed, the DynaPro NanoStar™ offers convenient cuvette-based DLS measurements over a very wide temperature range and with sample volume as small as 2 µL.
When accurate, high-resolution size distributions of nanoparticle suspensions are necessary, the analytical tool of choice is Field-Flow Fractionation (FFF) using an Eclipse™ FFF system, coupled to a DAWN™ MALS detector with an optional integrated WyattQELS™ DLS module. The NanoStar can also provide on-line DLS detection in conjunction with a Wyatt MALS detector, then easily be switched back to cuvette-based DLS.
FFF is conceptually related to size-exclusion chromatography, yet provides separation performance well beyond that of SEC, spanning the range of 1 – 1000 nm with excellent resolution. Whereas unfractionated DLS at best distinguishes particles that differ in size by 3-5x, FFF-MALS and FFF-DLS can characterize accurately particles that differ in size by just a few percent.
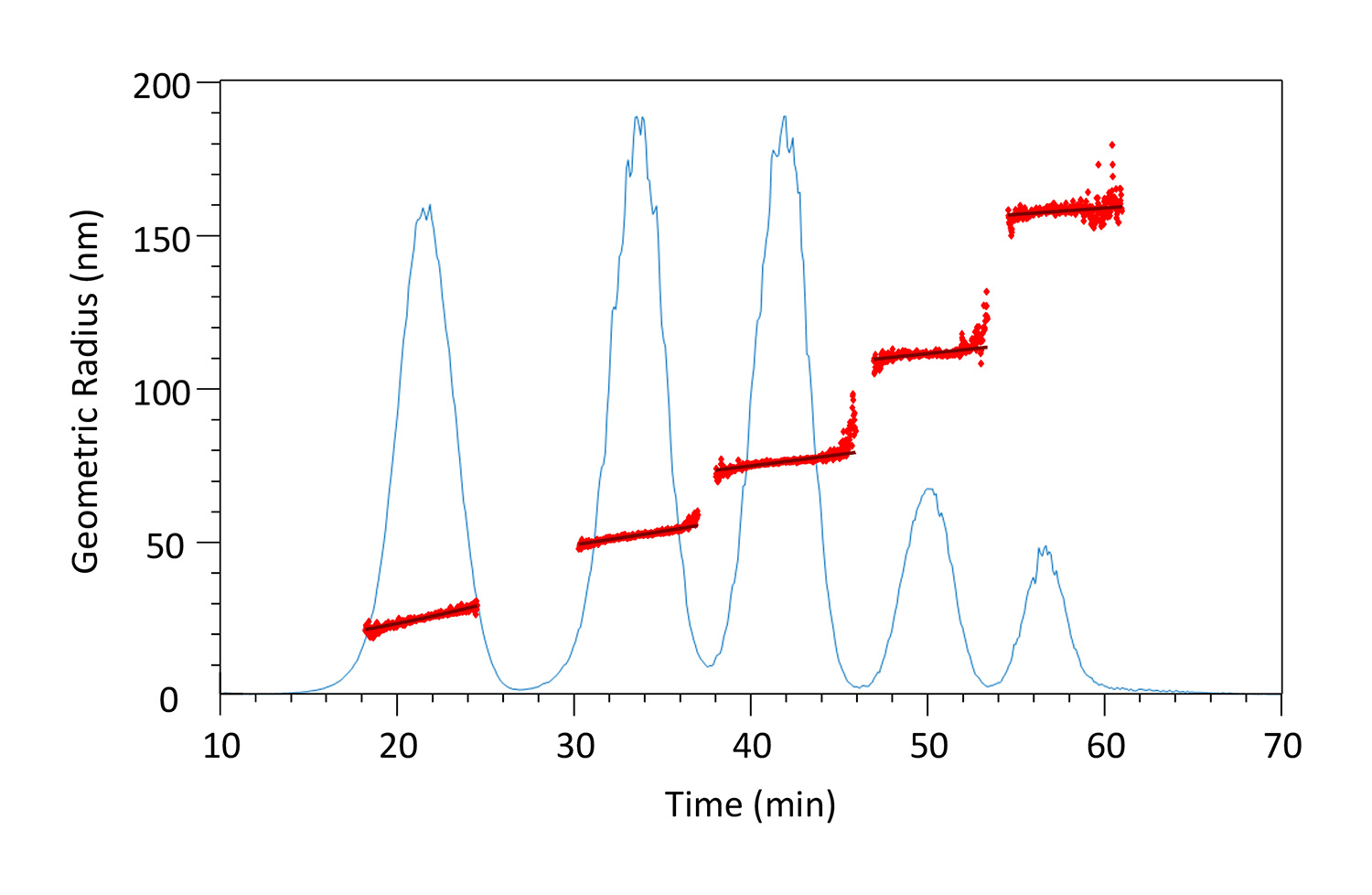
The DynaPro Plate Reader provides high-throughput, automated dynamic light scattering measurements, while the ZetaStar determines simultaneously both zeta potential and size by DLS.
High-resolution analysis of Duke particle standards by FFF-MALS shows the inherent polydispersity of each particle size.
Particle Concentration
Light scattering is an effective means of determining particle concentration (number if particles per mL) and quantitative particle size distributions. A relatively simple, quick and effective option for doing so is batch DLS in combination with single-angle static light scattering (SLS); this technique is most reliable when the sample comprises primarily one or two populations with distinct sizes, and the particle radius is no more than about 160 – 225 nm. NanoStar, ZetaStar and the DynaPro Plate Reader are capable of determining particle concentrations in this way.
A much more reliable and versatile method of determining particle concentration is FFF-MALS, which combines a size-based separation step with MALS detection and analysis. With knowledge of the refractive index of the particle material, MALS determines the numbers of nanoparticles in solution for each eluting size fraction. FFF-MALS analyzes the concentration and quantitative size distributions of viruses, liposomes, lipid nanoparticles, latex beads, extracellular vesicles and other nanoparticles with results similar to TEM counting to within a few percent. Particle concentration by FFF-MALS compares favorably with particle tracking analysis, covering a larger size range and without the need for careful dilution to within a narrow concentration range.
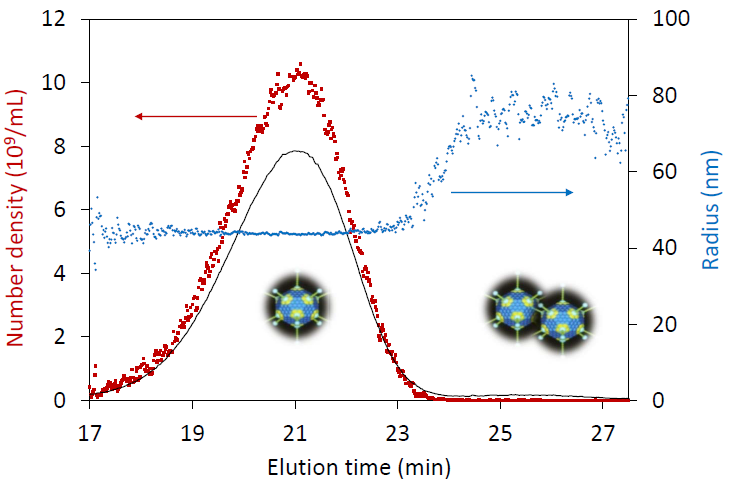
Analysis of adenovirus size and particle concentration for each eluting fraction by FFF-MALS.

An FFF-MALS system comprises Eclipse FFF controller and channel, DAWN MALS instrument, Optilab dRI detector along with pump, autosampler and UV detector.
Conformation
While light scattering does not provide high-resolution images of nanoparticles, it is an effective means of learning about the distributions of shapes or other conformational properties in particle ensembles. This is accomplished using three devices: an Eclipse FFF separation system, a DAWN MALS detector, and a WyattQELS DLS module integrated into the DAWN.
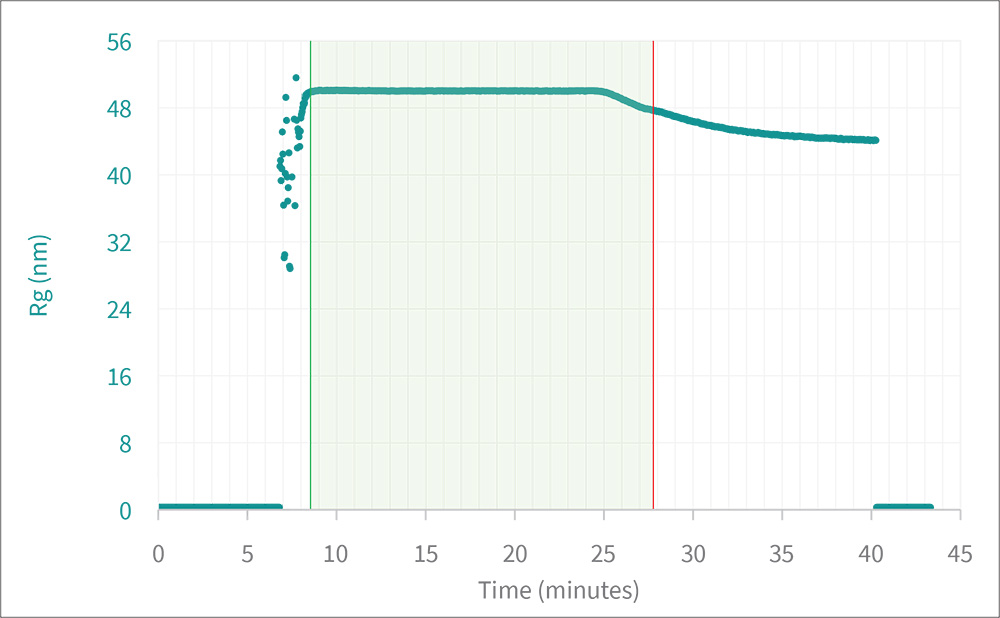
FFF separates the particles by size. As each elution volume enters the DAWN, light scattering makes two independent measurements: the rms radius Rg, by means of MALS, and the hydrodynamic radius Rh, by means of DLS. The ratio Rg:Rh, known as the 'shape factor,' provides an indication of a primary structural parameter.
Some a priori information about the expected shape is required for proper interpretation of the shape factor. If the particles are known to be elliptical and of uniform composition, the shape factor indicates the degree of deviation from sphericity and the axial ratio. If the particles are known to be spherical but may be hollow shells or uniform spheres, the shape factor indicates the actual conformation, and likewise for a variety of simple, well-defined structures.
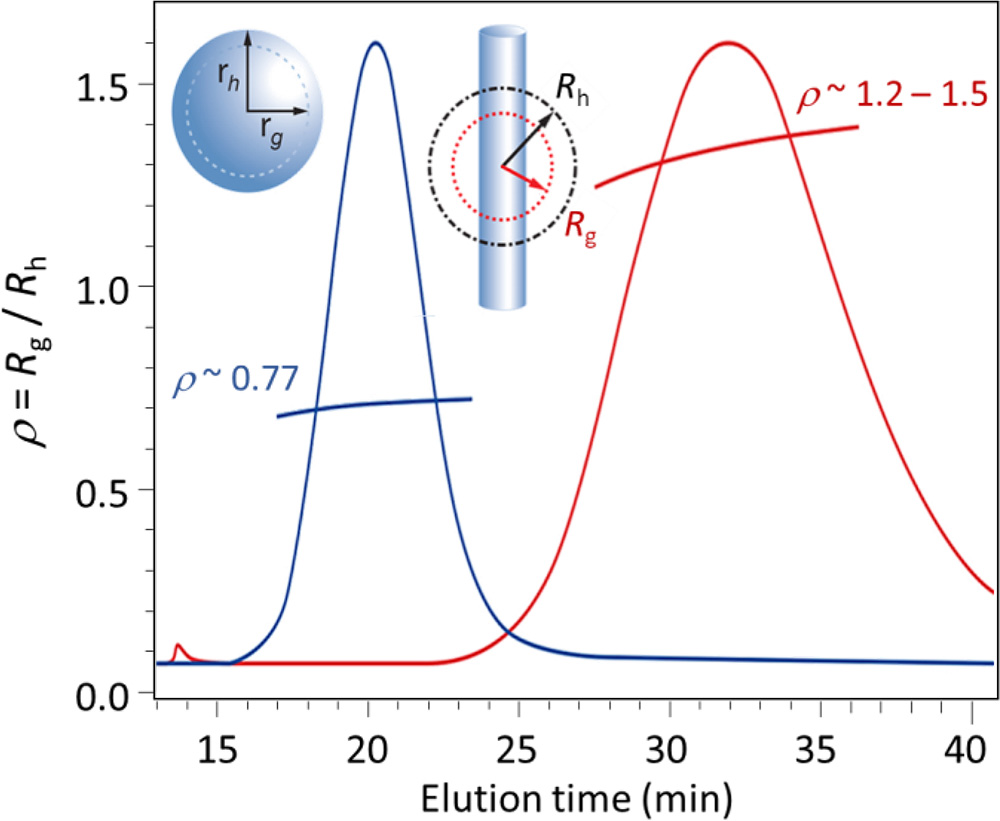
FFF-MALS-DLS shape factor analysis of two particles: spherical (expected value = 0.77) and rod-like (expected value > 1.1).
Composition
An FFF-MALS/DLS system can be further coupled to ICP-MS for elemental analysis. In this approach, the size and relative distribution of the eluting particles are correlated with elemental composition as confirmed by mass spectrometry. This combined technique has been used to examine the size and chemical composition of nanoparticles found in foods as well as the type of metals associated with nanoparticulate fractions recovered from natural sources such as lakes and streams.
The payload of nanodrug delivery particles such as lipid nanoparticles, liposomes or PLGA can be determined by SEC-MALS or FFF-MALS in combination with UV and RI detection. An example is provided in the controlled-release drug-delivery section on this page.
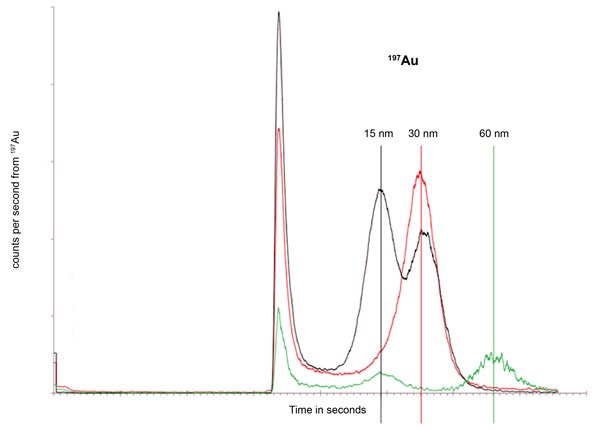
The gold content of particles of different sizes. Size determined by on-line DLS upstream of the ICP-MS system.
Techniques
Process Analytical Technology
In manufacturing processes of lipid nanoparticles, viruses, polymer micelles and other nanoparticles, most PAT instruments monitor process conditions such as pH, temperature, pressure and feedstock consumption. Only at the end of the process can QC procedures check the actual physical attributes that define product quality. Thanks to RT-MALS, this is no longer the case.
Real-time multi-angle light scattering (RT-MALS) is a novel PAT application of light scattering, that directly determines the size and concentration of nanoparticles with diameters from 25 to 500 nm. Using the ultraDAWN™ RT-MALS instrument, in-line or on-line with production, purification and other process steps, manufacturing staff can speed up and optimize process development with immediate feedback on the impact of process variations and deviations.
After developing the process, the ultraDAWN may be left on-line. Its accompanying software, OBSERVER™, integrates with an OPC-UA environment for automated and/or GMP operation. OBSERVER can be programmed to trigger the system to react to deviations from the allowed size range due to process drift, or to shut down a reaction once the endpoint has been reached.

Formulation
High-throughput screening of aggregation and stability of nanoparticles, in a plethora of permutations of solvents and solvent conditions, is essential for productive formulation efforts. The DynaPro Plate Reader is a true workhorse for formulators as it performs DLS screening on hundreds of samples, in situ and unattended, in standard microwell plates to assess size, polydispersity and aggregation. The DynaPro readily integrates with liquid handling robotics for even higher productivity.
If high-throughput is not needed, the cuvette-based DynaPro NanoStar is a versatile instrument which can also provide on-line DLS (e.g., FFF-DLS) when coupled to a Wyatt MALS detector.
In addition to DLS/SLS for size, molar mass, thermal and colloidal stability, the ZetaStar combines simultaneous DLS and ELS for size and zeta potential determination. Uniquely, zeta potential measurements in the ZetaStar may be made manually, or automatically by means of an autosampler using the flow cell. The Atlas™ accessory adds to the utility of the ZetaStar by enabling measurements even under conditions of high ionic strength. Like the NanoStar, the ZetaStar also couples to a Wyatt MALS instrument for online DLS measurements.
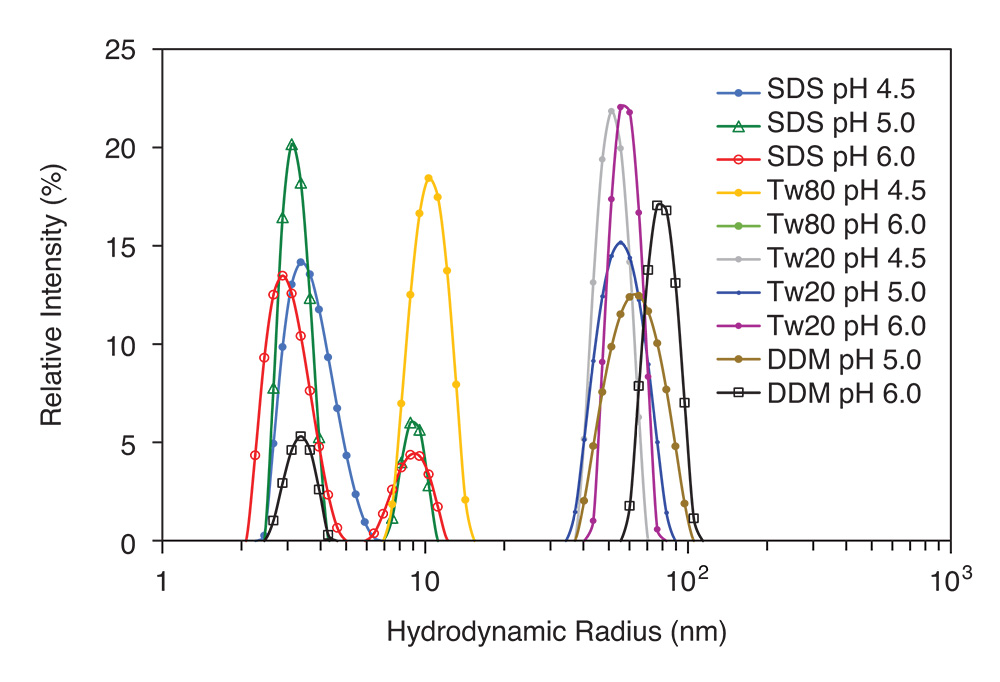
Automatically screen dozens or hundreds of nanoparticle formulations for size by DLS with a DynaPro Plate Reader.
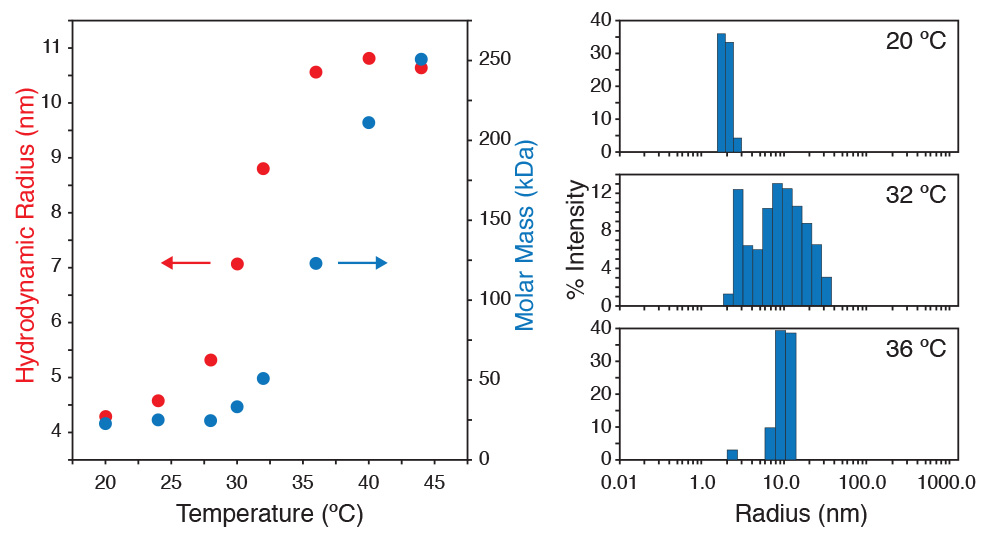
Simultaneous static and dynamic light scattering measurements in a DynaPro NanoStar cuvette-based DLS instrument reveal the critical micelle temperature of a polymersome. The NanoStar also couples optically to a DAWN MALS instrument to serve as an online DLS detector.
Nano-Formulation and Drug Delivery
Light scattering and field-flow fractionation provide characterization capabilities essential in developing novel nano-formulations, whether RNA-bearing lipid nanoparticles, drug-carrying liposome, or other carrier vehicles like PLGA micelles.
Basic sizing of nano-formulated particles is usually carried out initially in a batch (unfractionated) dynamic light scattering (DLS) instrument which makes measurements in a microcuvette, such as a DynaPro NanoStar or ZetaStar. Far more accurate size distributions are obtained using a separation step e.g. SEC-MALS for particles up to ~ 100 nm in size or SEC-MALS for particles up to 1000 nm in size. Both of these techniques are sensitive enough to resolve particles that differ by just a few nm in radius, amongst a heterogeneous distribution.
One of the most important analytical challenges in nano-formulation development is the determination of whether a drug, therapeutic peptide or nucleic acid is incorporated into the carrier, bound to the outside or free of the carrier. The combination of SEC or FFF with a DAWN MALS detector and WyattQELS online DLS module can help answer these questions by identifying the particle's conformation and content as a function of size, and also detecting and identifying unbound molecules.
For stable formulations, nano-formulated constructs may be evaluated in dozens or even hundreds of conditions in the DynaPro Plate Reader to minimize aggregation. The ZetaStar is ideal for measuring the zeta potential of drug-delivery nanoparticles in a series of formulation buffers, automated by an autosampler, as well as high-salt buffers that disrupt conventional zeta potential measurements when bubbles are formed at the electrodes by electrolysis. Zeta potential is an excellent indicator of the N/P ratios well as colloidal stability.
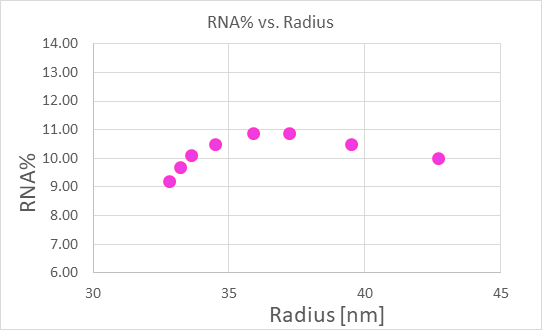
Nanoconjugate analysis of the RNA content versus size in an LNP formulation of mRNA, obtained with FFF-MALS-UV-dRI.
Resources
Application Notes
Selected References
Afonin, K. A.; Kasprzak, W.; Bindewald, E.; Puppala, P. S.; Diehl, A. R.; Hall, K. T.; Kim, T. J.; Zimmermann, M. T.; Jernigan, R. L.; Jaeger, L.; Shapiro, B. A. Computational and experimental characterization of RNA cubic nanoscaffolds. Methods 2014, 67, 256-265.
Dhayal, S. K.; Gruppen, H.; de Vries, R.; Wierenga, P. A. Controlled formation of protein nanoparticles by enzymatic cross-linking of α-lactalbumin with horseradish peroxidase. Food Hydrocolloid. 2014, 36, 53-59.
Loeschner, K.; Navratilova, J.; Legros, S.; Wagner, S.; Grombe, R.; Snell, J.; von der Kammer, F.; Larsen, E. H. Optimization and evaluation of asymmetric flow field-flow fractionation of silver nanoparticles. J. Chromatogr. A 2013, 1272, 116-125.
Perevyazko, I. Y.; Delaney, J. T.; Vollrath, A.; Pavlov, G. M.; Schubert, S.; Schubert, U. S. Examination and optimization of the self-assembly of biocompatible, polymeric nanoparticles by high-throughput nanoprecipitation. Soft Matter 2011, 7, 5030-5035.
Wyatt, P. Submicrometer particle sizing by multiangle light scattering following fractionation. J. Colloid Interf. Sci. 1998, 197, 9-20.