FFF-MALS
FFF-MALS: advanced separation and characterization of macromolecules and nanoparticles
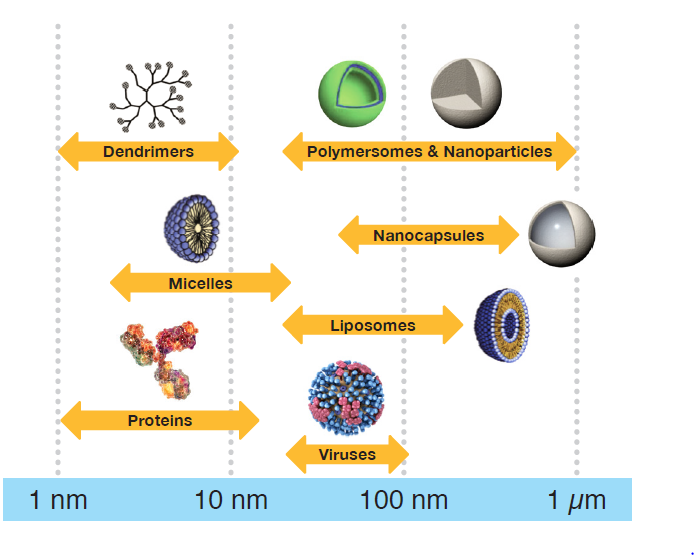
FFF-MALS is a powerful method to separate and characterize all types of analytes ranging in size from 1 nm to 1000 nm, such as:
- Gene vectors, viruses and nanodrug carriers
- Proteins, aggregates and assemblies
- High-molecular-weight as well as standard polymers
- Nanoplastics and metallic tracer nanoparticles
With FFF-MALS you can reliably determine high-resolution size distributions and other physical properties critical to the study, development, production and quality control of these denizens of the nano world.
What is FFF-MALS?
Field-flow fractionation (FFF) is a unique method for separating macromolecules and nanoparticles by size. The power of FFF resides in its essential tunability: simply by changing flow rates, a single separation channel can be used to explore complex samples comprising molecules, particles and emulsions across the entire size range, with superb resolution.
But separation is only half the battle. For comprehensive characterization, an Eclipse™ FFF system couples to the DAWN™ online multi-angle light scattering (MALS) instrument together with dynamic light scattering (DLS), refractive index and UV/Vis detectors to analyze:
- Size
- Molar mass
- Mass concentration
- Particle concentration
- Conformation
- Composition and particle payload
Clearly offering a much larger range than size-exclusion chromatography, FFF does share some similarities with SEC—both fractionate according to diffusion coefficient—but FFF uses no stationary phase. The analytes do not experience shear or high pressure. Beyond online analysis, size fractions can be collected for additional offline characterization.
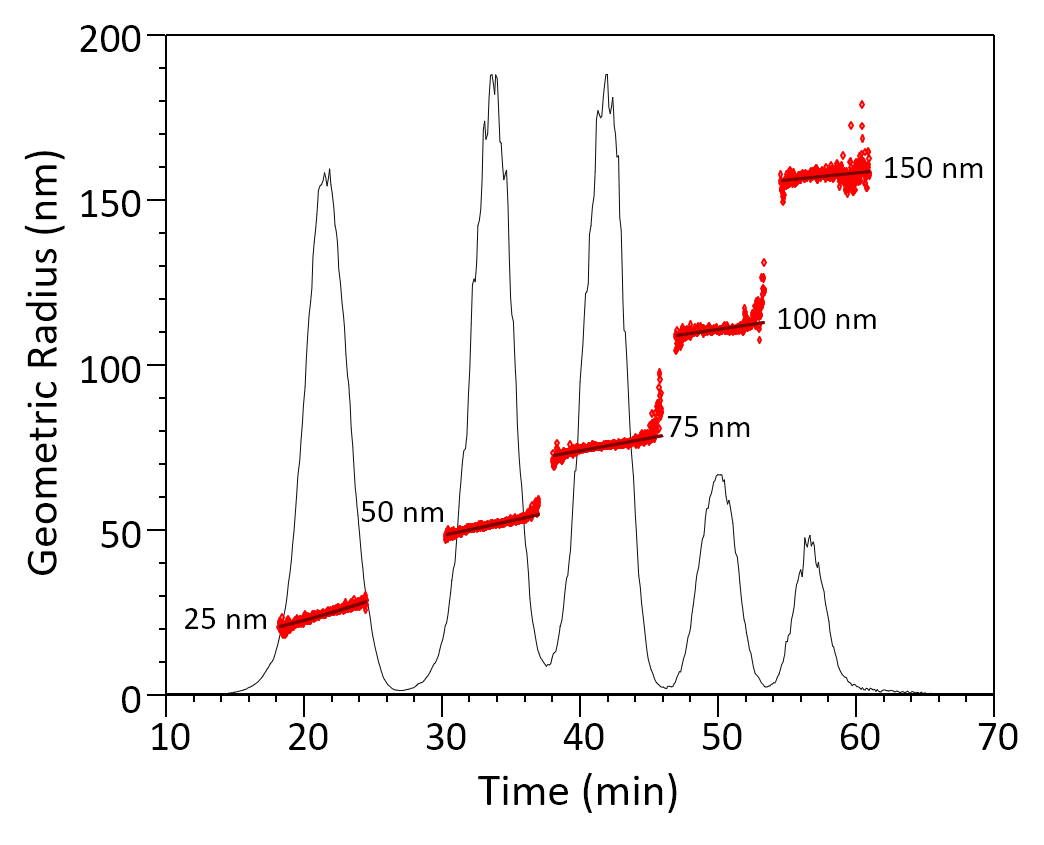
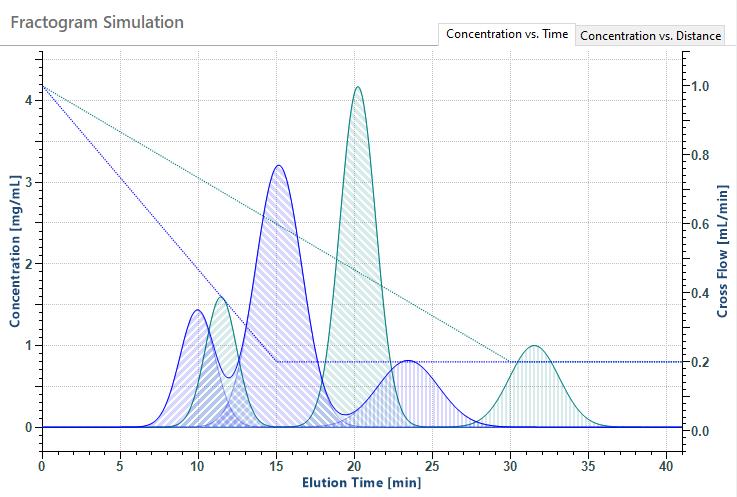
FFF separation of a mix or particles, illustrating excellent resolution. The average size and heterogeneity of each population is calculated from the sizes of all eluting fractions (slices) in the peak.
Why FFF-MALS?
Nanoparticles
FFF-MALS is the only analytical technique for nanoparticles that provides a complete set of advantages:
- High-resolution separation to enable detailed characterization
- A wide variety of online detection modes, including spectroscopic and multiple types of light scattering, for comprehensive characterization
- Non-destructive and capable of collecting fractions for further offline analysis, e.g. imaging
- Semi-preparative fractionation is also available. FFF is suitable for analytical and semi-prep scales from nanograms to milligrams
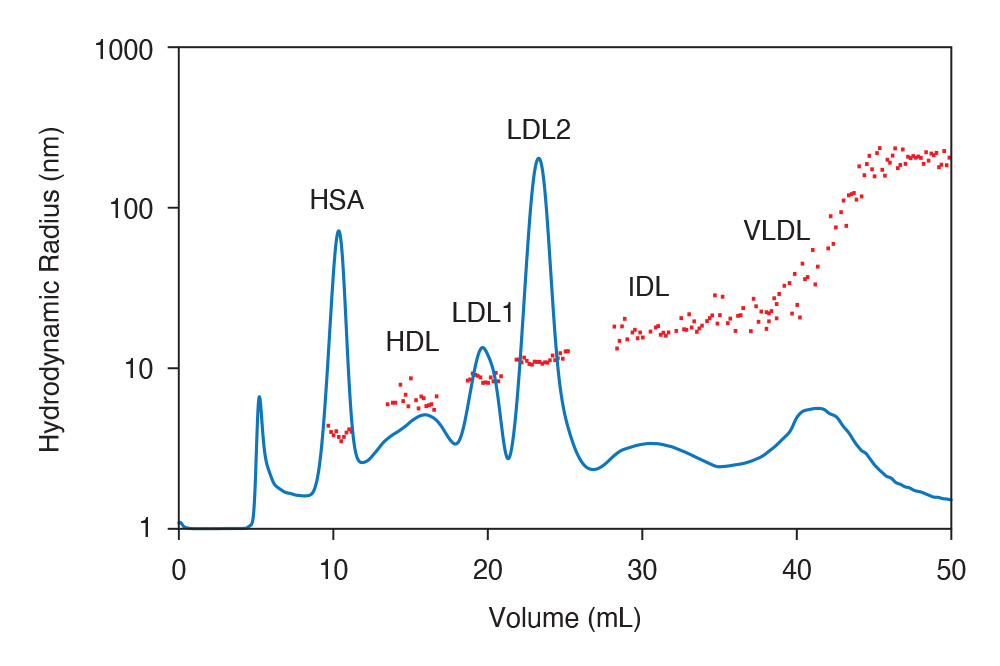
FFF-MALS-DLS separation of whole serum, with distinct peaks for serum albumin, IgG, and various types of lipoproteins with sizes ranging from 3 nm to 300 nm.
Macromolecules
For macromolecules such as HMW polymers and large protein aggregates, FFF-MALS is essential when the size range and complexity of the analyte exceed the capability of SEC. In such cases SEC-MALS is inappropriate for absolute characterization.
- Because FFF is not limited by pore size like SEC, its upper size limit for separation well exceeds that of size-exclusion columns.
- Large macromolecules and complexes are not filtered out, and with no packed column bed there is very little shear, so fragile structures are preserved.
- Anchoring effects, common in highly-branched HMW polymers, do not occur in FFF separation.
What’s more, the separation power, range and resolution can be tuned simply by adjusting the flow ratios, whereas a column has no adjustable parameters. That means that a single FFF channel can cover the range where SEC would require multiple columns.
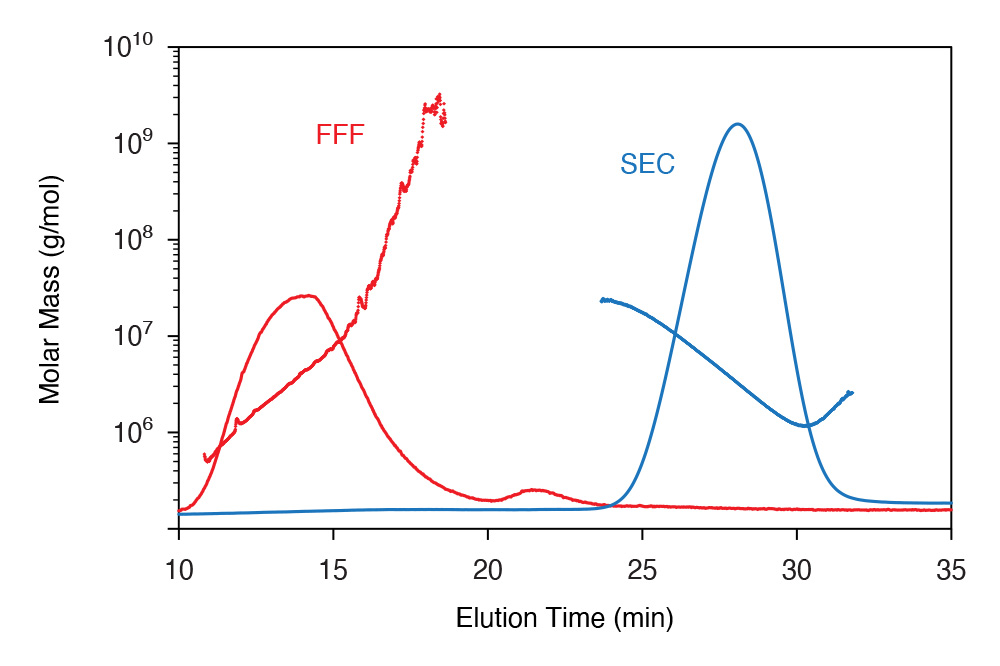
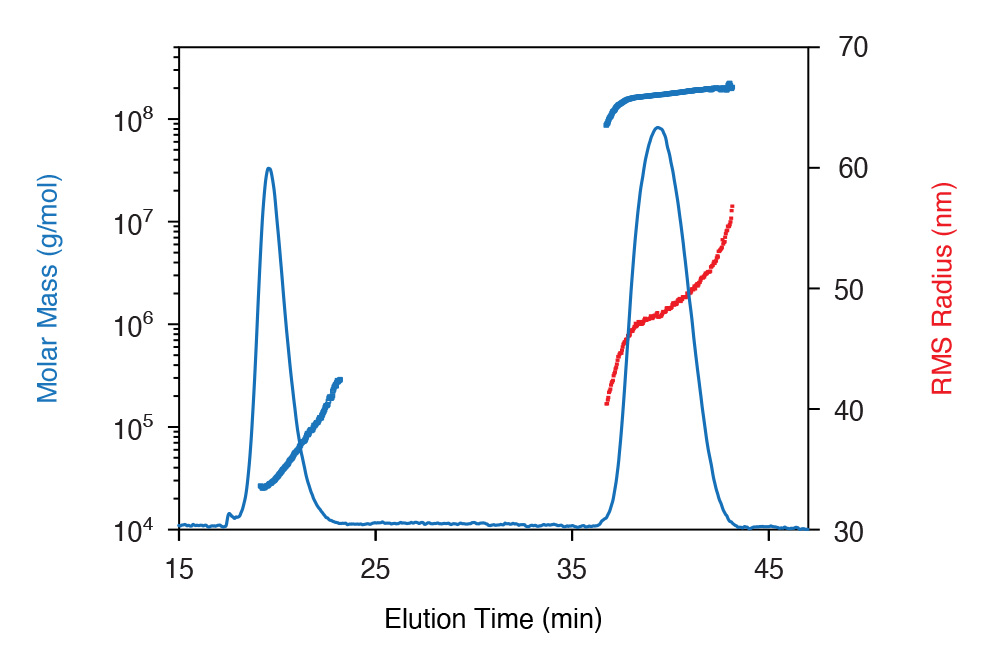
SEC and FFF separations of a high- molecular-weight protein-polysaccharide conjugate (PPC), spanning four orders of magnitude in MW. Fractions above ~107 g/mol were removed by the SEC columns. SEC separation also exhibited anchoring (upturn in MW on trailing edge). FFF provides near-ideal fractionation of this large conjugate for accurate MALS analysis.
Who uses FFF-MALS?
FFF-MALS is used to analyze complex samples with a wide size range, by:
- Biopharmaceutical scientists for characterization and process development of gene therapies, vaccines and biologics such as monoclonal antibodies
- Pharmaceutical technologists for developing nano drug-delivery systems such as liposomal formulations, nanocrystals and polymeric controlled-release particles
- Molecular biology and nanomedicine researchers for isolation of extracellular vesicles, novel drug and gene delivery studies
- Polymer analytical chemists for characterizing starches, hyaluronic acid, rubbers, lignins, cellulose and high-end latex particles
- Environmental scientists to study colloidal transport of nanoplastics, tracer particles and toxic materials in the environment
- Nanoparticle engineers for novel applications
- Food and beverage developers to understand and improve viscosity enhancers, food additives, proteins and biopolymers

The Eclipse/DAWN FFF-MALS system
FFF-MALS couples an Eclipse field-flow fractionation system, comprised of an Eclipse FFF instrument plus a pump and autosampler, with a DAWN multi-angle light scattering instrument and other online detector modules.
VISION™ software is used for all aspects of FFF while ASTRA™ software acquires and analyzes the detector signals. Both VISION and ASTRA are available as standard and 21 CFR Part 11 compliance options.

How does FFF-MALS work?
Separation in FFF-MALS is usually achieved by asymmetric-flow field-flow fractionation (AF4), the most prevalent form by far of field-flow fractionation (FFF). FFF utilizes an open channel, which gives it a large size range and versatility. Since the analyte separates according to hydrodynamic size, eluting fractions are generally quite homogeneous.
Following separation the eluting fractions are measured by one or more detectors including MALS, dynamic light scattering (DLS), UV/Vis absorbance, differential refractive index (dRI) and/or fluorescence.
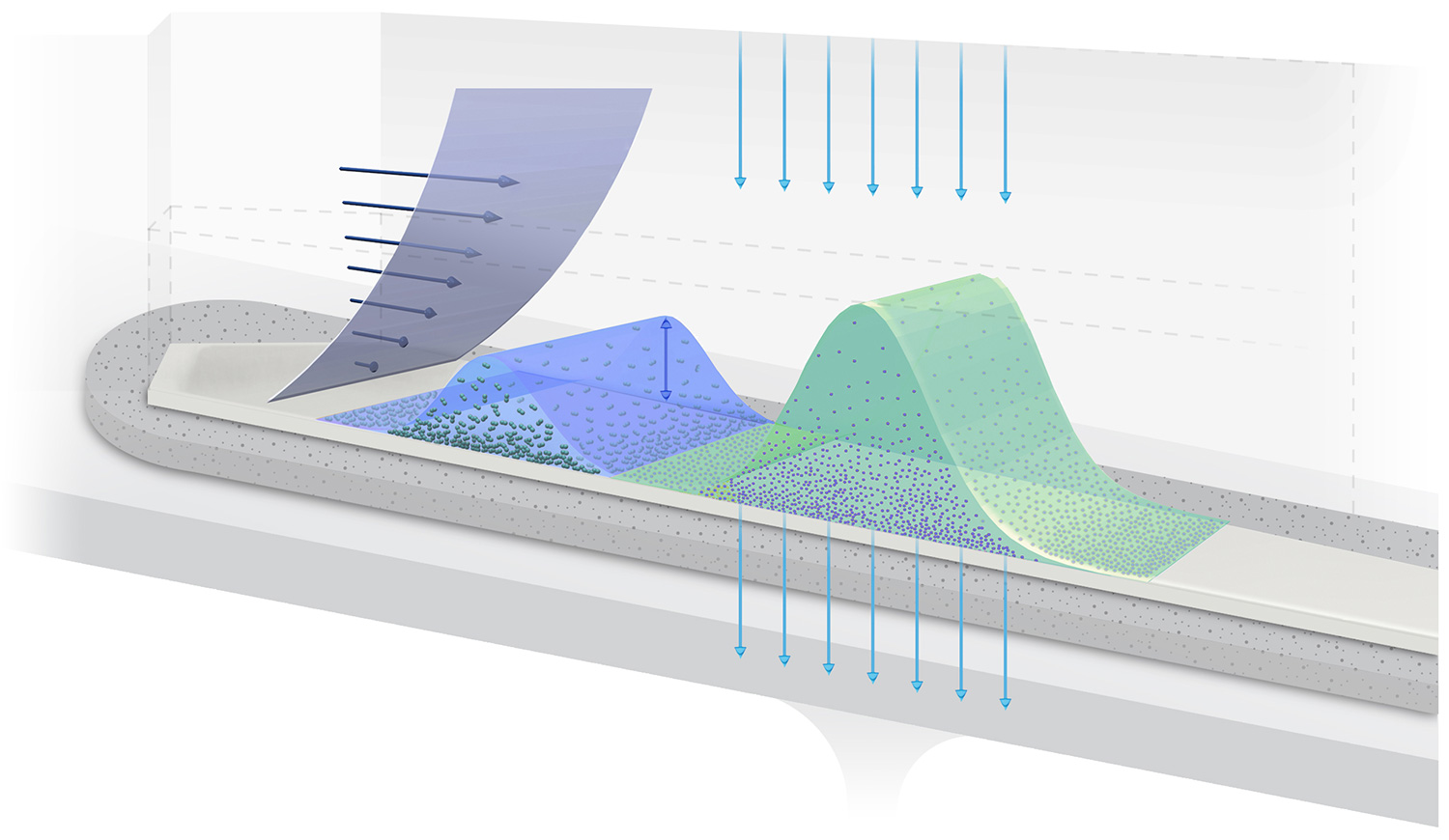
How FFF works
The principle of AF4 is illustrated in the image below. High-resolution separation is achieved in the thin, ribbon like channel by applying two flow streams: channel flow and cross flow.
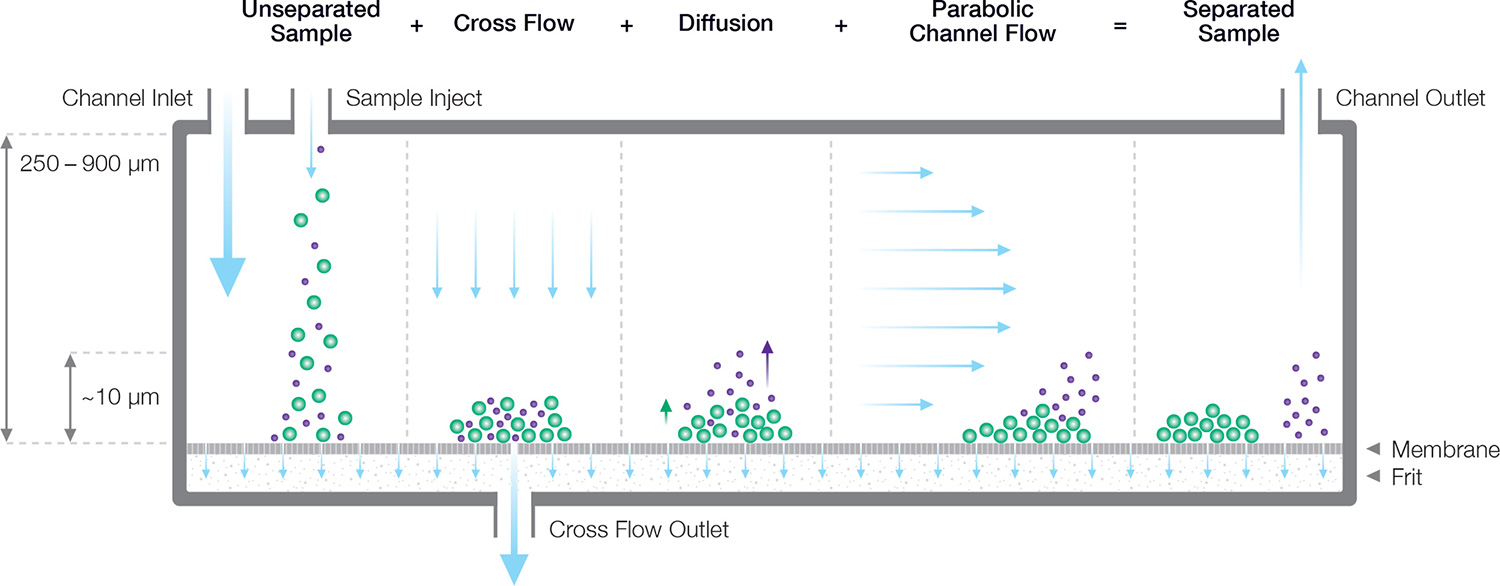
FFF separation is driven by particle diffusion working against a cross flow which concentrates them towards a membrane at the bottom of the channel. Smaller particles are on average higher up in the channel where they migrate faster in the higher velocity path lines.
Channel flow operates in parallel to the membrane and frit. Because the channel height is just a few hundreds of micrometers, the flow is laminar, resulting in a parabolic flow profile. The closer the sample is to the center of the channel, the faster it is swept out of the channel.
Cross flow operates perpendicularly to the semi-porous (ultrafiltration) membrane, which is selected to have smaller pores than the sample particle diameter, and is supported by the frit. The cross flow acts as a force field, concentrating the sample against the bottom wall.
Brownian motion represents a counter force working against the cross flow. Smaller particles diffuse more extensively against the cross flow, reaching further towards the center of the channel than larger particles.
Relaxation is the FFF step wherein equilibrium is established between cross flow and diffusion, leading to an exponential concentration profile up into the channel. The average height above the membrane depends on the diffusion coefficient of the sample.
During the elution step, different average heights of the sample components above the membrane results in different longitudinal transport velocities, towards the channel outlet. Smaller particles or macromolecules will elute first, followed by the larger components.
The principle of AF4 is illustrated in the image below or found in the video here. Additional details, an animated video of the FFF separation process, and information on electrical flow FFF and more may be found on the FFF Theory page.
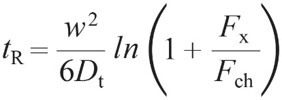
FFF separation under a constant ratio of cross flow to channel flow is described by a simple relation between retention time tR, channel height w, cross flow Fx, channel flow Fch and the particle’s diffusion coefficient Dt.
Electrical/Asymmetric-flow FFF (EAF4)
EAF4 is similar to standard AF4 with the addition of two electrodes. The application of voltage across the electrodes changes the particle distribution above the membrane according to the particle’s charge or zeta potential, and changes the retention time accordingly.
In order to analyze zeta potential, the shift in retention time is measured under at least two different applied electric fields (one of them is usually zero strength).
The principle of AF4 is illustrated in the image below or found in the video here. Additional details, an animated video of the FFF separation process, and information on electrical/asymmetric-flow FFF may be found on the FFF Theory page.
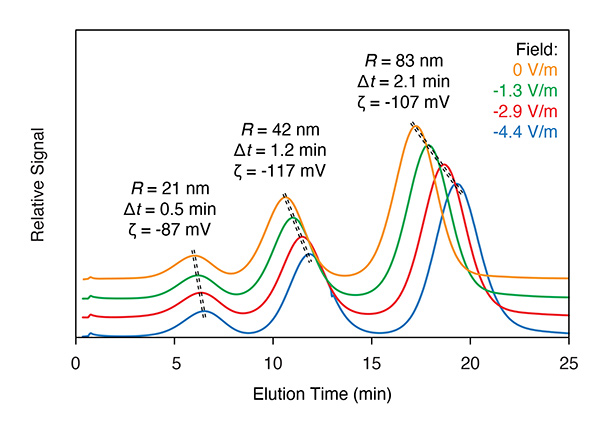
EAF4 determines the electrophoretic mobility and zeta potential of each particle size in a mixture.
MALS and other FFF detection technologies
Once separated, each size fraction may be characterized —independently of its specific retention time — using a variety of downstream detectors:
MALS
As each component passes though the detectors, its concentration and light scattering properties are measured every second or so. The MALS detector incorporates 18 photodiodes positioned at different angles θ relative to the laser beam in order to measure the scattered light. At each data point, the plot of scattered intensity vs. angle is fit to determine R(0) (the y-intersect at angle θ = 0) and the coefficients of an angular fit function.
- Macromolecules: Molar mass, M, is calculated from the ratio of R(0) and the concentration; radius of gyration, Rg, is calculated from the first-order term in the angular fit polynomial.
- Nanoparticles: While the angular data may also be fit to determine Rg for nanoparticles, it is more common to select a shape model such as a uniform sphere, coated sphere or rod for nanoparticles. These fits provide geometric dimensions such as radius or rod length. The particle concentration is calculated from R(0) and the particle dimensions, e.g. radius.
For more details on the theory of MALS, please see our MALS Theory page.
DLS
A dynamic light scattering detector measures the rate of fluctuation of the light scattering signal on time scales of microseconds and milliseconds. Since these fluctuations arise from Brownian motion, they can be analyzed to determine the diffusion coefficient and hydrodynamic radius Rh. Please see the DLS Theory page for additional details.
Compared to the usual batch DLS, FFF-DLS provides much higher resolution resulting from separation into homogeneous fractions measured in each time slice.
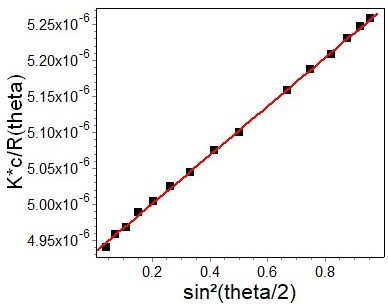
In MALS, scattered light is measured with detectors positioned at multiple angles around the analyte. The plot of scattered intensity vs. angle is fit to determine R(0) (the y-intersect at angle θ = 0) and the coefficients of an angular fit function. Molar mass and particle concentration are calculated from R(0), and size is calculated from the angular fit.
Additional detection modes
FFF-MALS makes use of additional detectors:
- A UV/Vis or differential refractive index (dRI) detector is used in conjunction with MALS to calculate molar mass of pure proteins or polymers.
- dRI may be combined with UV/Vis and MALS to analyze conjugated molecules such as glycoconjugates, protein-nucleic acid complexes or co-polymers.
- UV/Vis may be replaced by a fluorescence detector for labeled molecules at very low concentration.
- ICP-MS is used for elemental analysis of metallic and mineral nanoparticles; single-particle ICP-MS is used to count such particles while classifying them according to composition.
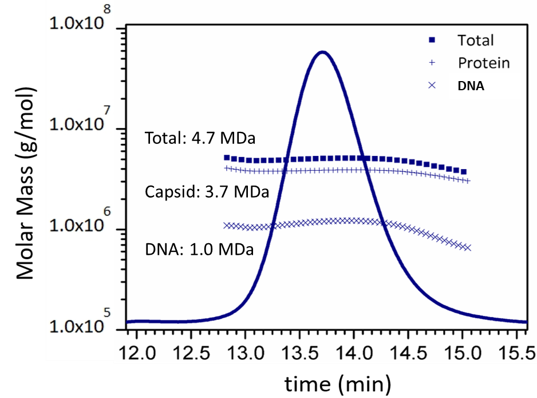
The combination of MALS, UV and dRI analyzes the content of conjugated macromolecules such as this AAV virus which comprises a protein capsid and DNA payload.
What constitutes a complete FFF-MALS system?
Basic
Separation system
The FFF part of FFF-MALS, which handles separations, consists of an Eclipse FFF instrument and separation channel, plus a single pump and an autosampler.
- The Eclipse instrument regulates channel, cross and injection flow rates. It also measures a variety of flows and pressures in order to continuously evaluate performance and provide feedback in case any issues should arise.
- The Short Channel is the most commonly used analytical FFF channel.
- An Agilent 1260 quaternary pump and autosampler serve as the liquid-handling front end. Other Agilent pumps and autosamplers are supported.
- VISION software controls the entire FFF system. VISION is also used for in silico method design and acquisition analysis of flow and UV signals.
Detection system
The basic FFF-MALS system includes a DAWN MALS instrument and either UV or dRI detector.
- DAWN has 18 angular detectors; it covers molar masses of 200 - 109 g/mol and radii of 10 - 500 nm.
- For analysis of polymers, lipid nanoparticles and other analytes that do not contain reliably known chromophores, an Optilab™ dRI detector is needed.
- For analysis of proteins and other UV-absorbing analytes including viruses, an Agilent 1260 VWD, MWD or DAD is included. Multi-wavelength data may be acquired from the MWD and DAD for additional analytics.
- ASTRA software acquires and analyzes MALS and concentration data as well as DLS data.
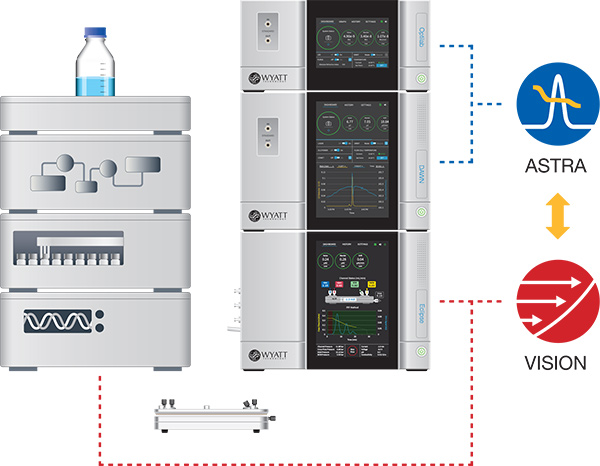
Advanced
Several advanced FFF options are available:
- SEC switching option: Automatically switches the entire system, including detectors, between an FFF channel and an SEC column.
- Dilution Control Module: The DCM reduces the amount of dilution of eluting sample between
1 - 10x, while providing extremely accurate detector flow rates. This improves sensitivity, repeatability, fraction collection and the DLS measurement range. - Eclipse Mobility: The Mobility add-on implements electrical flow FFF in order to measure the zeta potential of sample components.
- Additional channels:
- The Long Channel is useful in standard analytical FFF for increased sample load, and to reduce shear and membrane interactions via lower cross flow
- The Dispersion-Inlet Channel eliminates the focusing step for better separation of aggregation-prone samples
- The Semi-preparative Channel separates milligram quantities
- Fraction collector: An Agilent 1260 fraction collector may be added and controlled by VISION.
Additional detection modes may be added for even more comprehensive analysis:
- DLS module: Provides sizing of particles that cannot be measured by MALS: radius below 10 nm, and metallic nanoparticles below ~ 100 nm. The combination of MALS and DLS informs as to particle shape and structure. DLS may be added as a WyattQELS™ module embedded in the DAWN, or as a DynaPro™ NanoStar™ or ZetaStar™ stand-alone instrument connected via optical fiber to the DAWN flow cell.
- Dual concentration detectors: The combination of UV plus dRI is used to determine the content of conjugated molecules such as protein-polysaccharide conjugates or viral vectors.
- Fluorescence: An Agilent 1260 fluorescence detector is useful in identifying particles according to chemical composition or labels, and in detecting trace amounts of certain molecules.
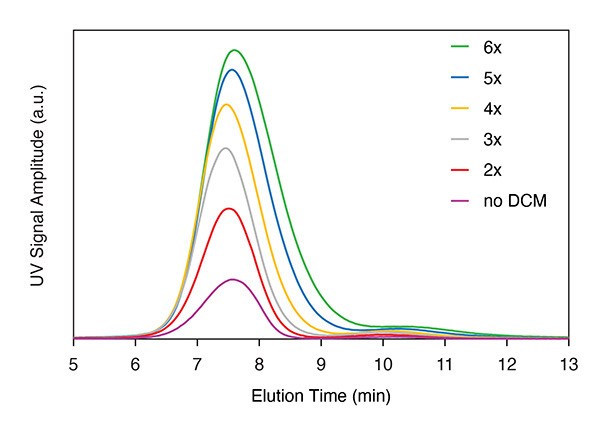
The Dilution Control Module enhances concentration at the detector up to 10-fold, and up to 5-fold without significant loss of resolution. Shown here: BSA monomer and dimer.

What is unique about the Eclipse FFF-MALS system?
- Performance: Best in the market with respect to resolution, sensitivity and size range of the separation
- Results: Powerful, comprehensive analyses of MALS and DLS data
- Software-aided method development: Eliminates laborious trial-and-error method development
- Ease of use: Start a sequences with one mouse click. Everything else, including system monitoring and data collection are fully automated.
- Productivity: System monitors and health indicators make sure every sample run produces optimal data. Reduced down-time with preventive maintenance reminders.
- Intelligent solvent management: Reduces buffer consumption so you can run more samples over extended sequences
Key FFF-MALS applications
FFF-MALS is used in many applications, in industry and research institutions. Every type of complex sample in solution or suspension in the submicron range can be analyzed with FFF-MALS. Below you will find a select set of application notes and on-demand webinars for each area. The searchable Bibliography, containing thousands of references to peer-reviewed articles citing Wyatt instruments, is an excellent resource for researching specific FFF-MALS applications.
Vaccines, gene vectors and extracellular vesicles
Characterization of gene vectors
Multiple characteristics of gene vectors are quantified by MALS coupled to SEC and FFF: identity, purity, homogeneity, nucleic acid content, particle concentration, empty:full ratio and more. The range and depth of such characterization is rapidly expanding.
Learn how FFF-MALS along with SEC-MALS and DLS quantify gene vector critical quality attributes by viewing the webinar Quantifying Viral Vector Attributes with Light Scattering.
Small viral vectors and virus-like particles
Small viral vectors and VLPs can be separated and analyzed by SEC-MALS to assess identity, content, particle concentration and aggregation. However, some VLPs bind to SEC columns, requiring a different separation technique – FFF fulfills the requirements. In addition, large aggregates may be filtered out or disrupted by the column, requiring a technique such as FFF which does not filter or shear these species.
Extended characterization of AAV aggregates is reported in AN2003: Quantifying AAV aggregation and CQAs by FFF-MALS.
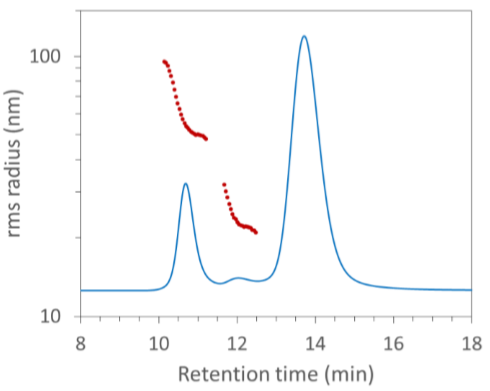
FFF-MALS analysis of AAV aggregation, from AN2003
Extracellular vesicles - isolation and characterization
While accurate, high-resolution size distributions obtained with FFF-MALS-DLS are important in EV research, the ability to isolate EVs from impurities and collect size-based fractions for offline analysis is invaluable. Analysis of such fractions lead to the discovery of three essential exosome populations: large exosomes, small exosomes and exomeres.
An introduction to EV separation, characterization and isolation is provided in WP2606: Exosome-characterization-by-FFF-MALS-DLS.
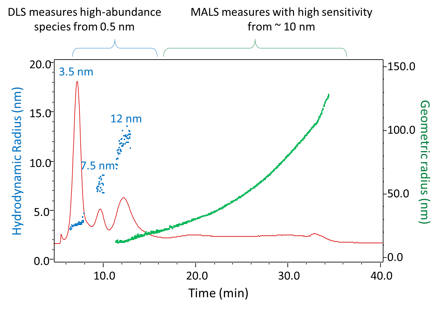
FFF-MALS-DLS analysis of extracellular vesicles, from WP2606
Characterization of vaccines
FFF-MALS and other light scattering techniques are used extensively for the characterization of vaccine agents such as viruses, virus-like particles, mRNA or DNA vectors, polysaccharides, and protein-polysaccharide conjugates.
A list of key publications on this topic is available as PL9006: Key publications on the characterization of vaccines with multi-angle and dynamic light scattering. This topic is also reviewed in the webinar Vaccines Illuminated: Biophysical characterization, PAT, and quality control via light scattering techniques.
Peer-reviewed publications
FFF-MALS is deemed essential for the characterization of viral and non-viral gene vectors.
A list of key publications on this topic is available as PL2609: FFF-MALS-DLS nanoDDS key publications.

Nanopharmaceuticals
Introduction to characterization of nanopharmaceuticals by FFF-MALS
FFF-MALS is a powerful technique for nanopharmaceutical characterization, in that it can quantify detailed size distributions, particle concentration, shape and content.
The principles of FFF-MALS for separation and characterization of nanopharmaceuticals are reviewed in WP2611: Characterization of nano-pharmaceuticals with field-flow fractionation and light scattering (FFF-MALS-DLS).
Methods for FFF-MALS characterization of nanomedicines, developed by NCI-NCL and the EU-NCL, are available at NCL Method PCC-19: Asymmetric-Flow Field-Flow Fractionation and EUNCL-PCC-22: FFF-MALS method development and measurements of size and molecular weight.
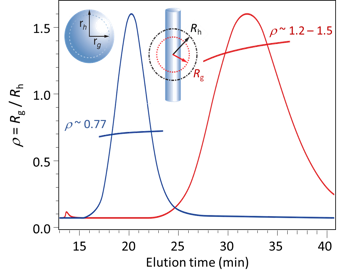
FFF-MALS analysis of particle shape is discussed in WP2611.
Lipid nanoparticle drug formulations
One of the primary challenges in developing and commercializing novel nanoparticle-based therapies is detailed characterization of the formulation components. FFF-DLS-MALS technology, which couples light scattering and other online detectors to field-flow fractionation, addresses this challenge, even in serum. The technique is suitable for meeting FDA requirements for analysis of liposomal formulations.
View the webinar Characterization of Lipid Nanoparticle Drug Formulations for case studies that include measurement of detailed liposome size distributions, assessment of drug release and transfer to cell membrane proxies; particle morphology, quantification of co-existing colloidal structures; and stability of the nanocarriers.
Polymersomes for drug delivery
The ability to characterize polymers and polymersome architectures, as well as their performance in terms of drug encapsulation, is critical in developing novel drug-delivery modalities that are carefully tailored to the therapeutic task.
Learn how FFF-MALS quantifies free:bound drug as well as polymer architecture and polymersome size distributions in the webinar Separation and Characterization of Complex (Bio)macromolecular Architectures.
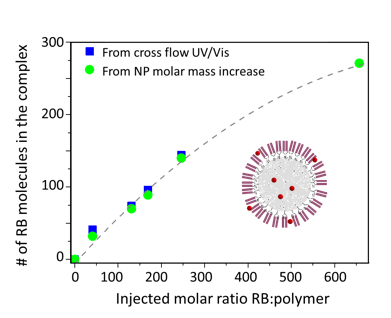
FFF-MALS analysis of small-molecule encapsulation, from WP2611. Figure courtesy Albena Lederer.
Peer-reviewed publications
FFF-MALS is deemed essential for the characterization of nanoformulated drugs.
A list of key publications on this topic is available as PL2609: FFF-MALS-DLS nanoDDS key publications.
Proteins
Protein aggregates
Light scattering offers a suite of complementary techniques for characterizing the irreversible and reversible aggregation of therapeutic proteins such as monoclonal antibodies. These include FFF-MALS as well as SEC-MALS, CG-MALS and dynamic light scattering.
Learn how these techniques provide a complete picture of mAb aggregation in the poster Aggregate Analyses by Light Scattering – Complementary and Orthogonal.
Protein-polysaccharide conjugates
Size analysis of complex, heterogeneous samples such as protein-polysaccharide conjugate vaccines can be quite challenging. Complete characterization requires not just molar mass and size distributions but also conjugation ratios.
An FFF-MALS method was developed for a 58 kDa protein coupled to polysaccharides in the range of 200 – 500 kDa, and the results compared to other techniques, in the poster Vaccines: Protein-Polysaccharide Conjugates.
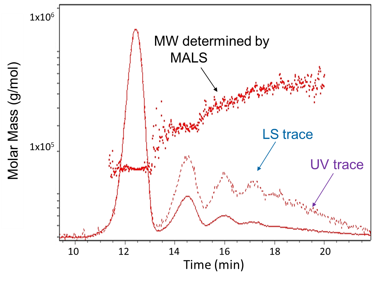
FFF-MALS analysis of protein aggregates.
Host-guest chemistry
Host-guest chemistry conjugates a hollow compound (host) via non-covalent interactions to one or more molecules displaying recognition moieties (guests). The formed complex can present stability issues during characterization analyses.
The ability of FFF-MALS to maintain high concentration during separation, thus avoiding dissociation of the conjugate and measuring the complex stoichiometry, is presented in AN2001: Stoichiometry and Stability of Host-Guest Protein-Peptide Complexes.

Host-guest system characterized in AN2001.
Cross-linked proteins
Enzymatic cross-linking of milk proteins enhances the texture and physical properties of fermented dairy products. Size and molecular shape play a major role in the formation of optimal gel networks, but size determination by standard separation techniques such as size-exclusion chromatography or gel electrophoresis is very challenging.
The use of FFF-MALS to separate and characterize cross-linked casein for molar mass and conformation is explained in AN2002: Enzymatically Cross-linked Casein Characterized by FFF-MALS.

Cross-linking time dependence of casein conformation from AN2002.
Biopolymers and synthetic polymers
Absolute analysis of molar mass, size and branching
While conventional calibration for gel permeation (GPC) or size-exclusion chromatography (SEC) is useful, there are inherent disadvantages in this type of analysis that introduce experimental error. Multi-angle light scattering (MALS) detection is quite simple to use and can help overcome the challenges faced with conventional calibration-based methods.
Learn about the fundamentals of MALS and its application to the characterization of polymers in the e-book Beyond GPC: Light Scattering for Absolute Polymer Characterization.

Branching analysis
Long-chain branching is one of the most important characteristics that impacts the material performance of polymers. SEC-MALS and SEC-MALS-IV are most appropriate for small and intermediate polymers, while FFF-MALS provides superior separation and characterization of large and highly branched macromolecules.
The theory and applications of light scattering and differential viscometry in branching analysis are examined in WP1003: Branching revealed: Characterizing molecular structure in synthetic polymers by multi-angle light scattering and in the webinar Branching Revealed: Characterizing Molecular Structure in Synthetic and Natural Polymers by Multi-Angle Light Scattering.
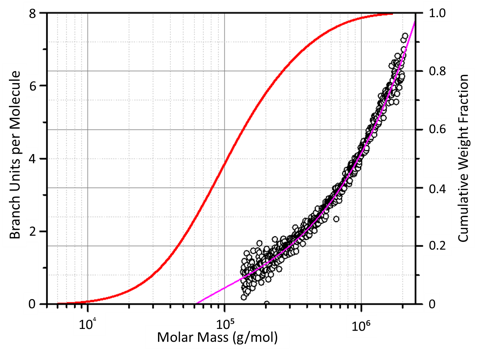
Polymer branching analysis from WP1003.
Lignin and lignosulfonate
Lignin and lignosulfonate analysis are among the most demanding applications of SEC-MALS and FFF-MALS. Their absolute molar mass distributions are characterized successfully via FFF-MALS, aided by the advanced technical features of the DAWN and the Eclipse.
Applications of MALS for lignin and lignosulfonate are reviewed in WP2303: Lignin and lignosulfonate characterization with SEC-MALS and FFF-MALS while applications to lignocellulosic material are explored in the webinar Pulp Non-Fiction: Absolute Macromolecular and Nanoparticle Characterization of Lignocellulosic Materials.
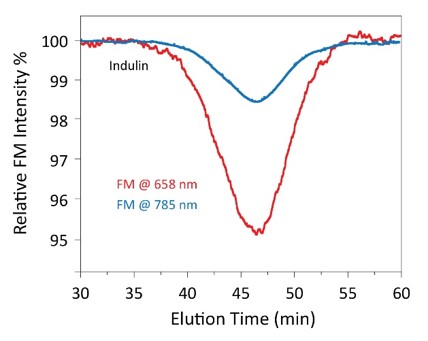
MALS laser absorption by lignin is quantified by the forward monitor, from WP1003.
Industrial lignosulfonate analysis
Lignosulfonates hold great promise for renewable materials using a common, inexpensive byproduct of pulping processes. Analysis of technical lignosulfonates by GPC requires extensive and laborious sample preparation.
FFF-MALS inherently removes impurities from lignosulfonates, enabling a single-step process for purification and characterization, as described in the application note AN2301: Lignosulfonate analysis by AF4-MALS.
Analysis of polymers in organic solvents
Numerous properties of technically important polymers utilized in the paint, rubber and adhesive industries are dependent on details of the molar mass distribution and molecular structure. Polymer chain branching, in particular, is a key determinant of many of these properties.
The analysis of synthetic polymers by FFF-MALS in organic carrier solvents is reviewed in the white paper WP2304: Analysis of polymers in organic solvents by FFF-MALS.
FFF-MALS characterizes both dissolved molecules and suspended nanogels in a single run, from WP2304.
Engineered and environmental nanoparticles
Single-particle ICP-MS
Engineered nanoparticles (ENPs) are coming into widespread use and so have become the subject of scientific interest in terms of possible toxic effects on humans and the environment. One key challenge is detection and identification of metallic ENPs in a complex fluid matrix. Inductively-coupled plasma mass spectrometry is extremely sensitive and can even be configured to identify and size individual particles.
ICP-MS and sp-ICP-MS were coupled to an Eclipse FFF system to monitor changes of particle size distribution and recovery caused by variation of ionic strength in AN2604: Characterization of Gold Nanoparticles with AF4-ICP-MS.

Environmental tracers
The combination of FFF-MALS-DLS with ICP-MS has become important in environmental analysis to evaluate the composition, size and number of particles present in solution. This technique can identify the size and elemental composition of particles at the parts-per-trillion level.
Examples of FFF-ICP-MS used to identify tracer nanoparticles in soil samples originating from wastewater and landfills are shown in the webinar Mass, Size, Composition: Field-Flow Fractionation of Biomolecules and Nanoparticles plus Characterization by Light Scattering and ICP-MS.
Silver-nanolipid complexes
Silver, especially microsilver, shows anti-microbial activity due to the oligodynamic effect. The combination of silver and nanostructured lipid carriers (NLC) leads to the formation of a silver–nanolipid complex. This complex, developed as a cosmetic product for sensitive skin, is also active against atopic dermatitis. However, in formulation work, standard methods such as DLS and zeta potential cannot confirm adsorption of microsilver to the surface of the NLC.
FFF-MALS proved to be highly sensitive to formation of silver-nanolipid complexes, as shown in the poster Silver Nanolipids.
Eclipse-VISION-ASTRA: A seamless workflow
With built-in intelligence and sophisticated software, the Eclipse system innovates and simplifies the complete workflow of FFF-MALS experiments. This system makes the full power and versatility of FFF-MALS accessible to technicians and occasional users as well as expert scientists.

How do I use FFF-MALS?
The Eclipse FFF system is designed for a streamlined and fully automated workflow, making it straightforward to develop, optimize and run a separation method. Once a method has been established, it is just a matter of pipetting samples into the autosampler vial and clicking ‘Run’. Eclipse™, DAWN™ and VISION™ will let you know if everything is on track or needs your attention.

Computer-aided method development
Let your PC do the work
The versatility and flexibility of FFF—where the separation power, range and sensitivity depend on flow rate ratios—means that an optimized flow program is usually developed for new samples. The VISION DESIGN module carries out physics-based FFF simulations for in silico method design and optimization, so there is no need to run a matrix of test methods.
Just list the estimated particle sizes, select your FFF channel, membrane and spacer, and specify a cross-flow gradient. VISION DESIGN immediately displays the predicted fractogram using fundamental FFF theory to calculate retention times.
It only takes a few minutes to explore the effect of different cross-flow rates and timings to improve resolution and find the best method - all from the comfort of your desk.
Refine the method
The final design can be transferred in a click to VISION RUN to test your method. Your initial guess of sample and channel parameters may have been inaccurate, but have no concern—results of a physical experiment can be fed back into VISION DESIGN to further refine the method. Usually a single test run suffices to give VISION DESIGN the information it needs to help you fully optimize your method.
Sample and system preparation
Light scattering is especially sensitive to particulates, which are usually the main source of noise in the MALS signal but are almost entirely absent from UV and dRI signals. Eliminating particles from sample and solvents is not difficult, but it does require some diligence.
Samples should be filtered with pore sizes of 0.22, 0.45 or 1 µm per the expected size range. Mobile phase should be filtered to 0.1 µm, though acceptable results might be obtained with the more common 0.22 µm filters. In addition, a 0.1 µm filter is added to the system between the pump and injection loop.
Is my system clean?
After flushing the system with a new solvent, it may not be obvious if the system is sufficiently particle-free for high-quality MALS measurements. The System Status Monitor on the front panel of the DAWN let you know if the noise level is below the recommended maximum for acceptable data.
Are the fluidics working properly?
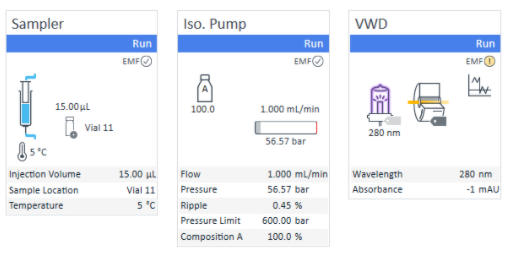
Steady, ripple-free flows are important for the best resolution and repeatability in FFF separations. VISION RUN lets you know if they are within acceptable levels. The Eclipse front panel displays flow rates and pressures, and will let you know if any of the pressures exceed limits.
All systems go?
Method status is evident with a glance to the Eclipse display. Continuous self-diagnostics in the Eclipse and DAWN ensure that you will know right away in the event of any conditions leading to sub-optimal runs. You will also receive actionable advice on how to return the system to top performance. Maintenance monitors will inform you when it is time to perform preventative maintenance.
System health indicators on the DAWN
System status displayed on the Eclipse.
Running an FFF-MALS experiment
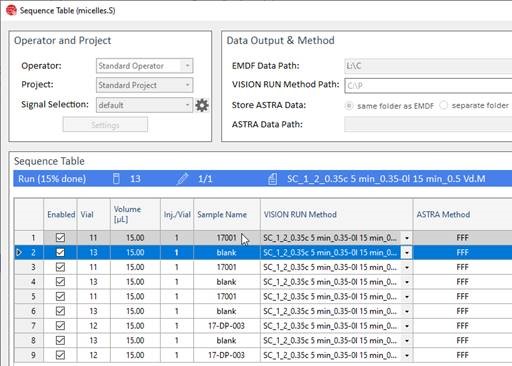
After pipetting samples into the autosampler vials and a priming a bottle of carrier liquid to the pump, the sequence is set up in the VISION RUN module using the method(s) developed in VISION DESIGN or established method(s). The entire set of samples is then run at the click of a mouse.
Enter the cockpit
VISION RUN is a sophisticated control center for the Eclipse FFF system. The dashboard shows the system status in real time with direct access to instrument configurations and sample sequences. Status information from the Eclipse instrument’s Health Indicators are displayed; should any warnings come up, you will receive actionable recommendations. New samples and additional injections may be added while running a sequence.
Expand your horizons
VISION RUN controls Agilent® modules from the 1260 series. The complete feature sets of autosamplers, isocratic or quaternary pumps, diode array detectors, fluorescence detectors and fraction collectors are supported to extract the maximum from FFF experiments.
Look ahead, see more
VISION RUN synchronizes and launches ASTRA software, which is responsible for data acquisition and analysis from the light scattering, RI and UV detectors. While ASTRA operates in the background to acquire data, the signals may be viewed in VISION.
Separately, VISION RUN acquires UV and flow data for diagnostics and method refinement; it also acquires pH and conductivity measurements if the Mobility EAF4 module is installed.
Data analysis
Analysis of detector data for sample characterization is usually carried out in ASTRA™ software. In certain circumstances, Eclipse and UV data are imported back into VISION for additional analysis.
MALS, DLS and more
Processing of light scattering, UV and RI data to determine molar mass, size, composition and conformation is done in ASTRA, which is laid out like standard chromatography software. Enter a few samples-specific parameters such as UV extinction coefficient and dn/dc, then set baselines and select peaks and the software does the rest. The results for multiple runs can be overlaid in EASI Graph and collated in EASI Table.
Details on the various analyses and reporting capabilities in ASTRA may be found here: https://www.wyatt.com/products/software/astra.html.
Particle size, method refinement and zeta potential
Flow, UV and electrical data recorded in VISION may be processed in VISION for specific analyses:
- Estimates of particle size, obtained from the FFF retention volume
- Refining the separation method, since the particle sizes determined may differ from the initial guess as to the sample content
- Calculation of zeta potential by measuring the shift in retention of each peak under two or more electric fields
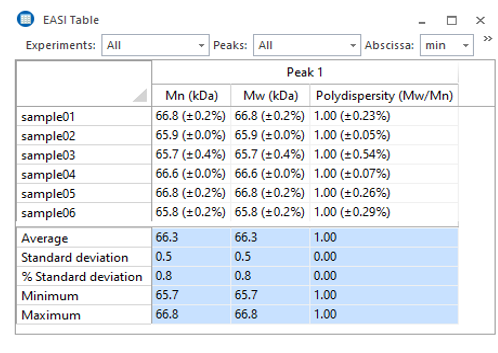
ASTRA’s EASI Table consolidates results from multiple data files.
System maintenance
Maintenance of the Eclipse/DAWN FFF-MALS system is minimal. The pump filter membrane is usually changed once every month or two, and ultrafiltration membrane in the FFF channel every ~ 100 runs. The system can be cleaned and sterilized with 20% ethanol or a standard detergent flush. Service reminders and an extensive log of system parameters help to maintain the system in perfect shape and prevent down time.

The modules that make up an FFF-MALS system
An Eclipse/DAWN FFF-MALS system comprises only the most advanced and reliable component sub-systems. The primary modules are designed and manufactured in the USA by Wyatt Technology, and other components are top-of-the-line Agilent products. The entire system is supplied, installed and serviced by Wyatt Technology.*
* In markets not served by Wyatt Technology field offices, Agilent products may be supplied and serviced by local Agilent distributors.

FFF components
Eclipse™
The Eclipse FFF instrument is the heart of the FFF system. It incorporates four highly precise and stable flow controllers that ensure the most robust and repeatable fractionation runs, without the associated pulsation and inherent unreliability of the multiple pumps in other FFF systems.
Eclipse presents an intelligent user interface that reflects its sophistication, and informs the user of events or conditions that need attention. In addition, this design combines tip injection and focus-zone injection, enabling use of a variety of FFF separation channels and even a size-exclusion chromatography column.
Optional features:
- FFF-SEC switching: This option supports two different separation channels, usually one FFF channel plus an SEC column, sharing the same autosampler, pump and detectors. At the click of a mouse you can change your separation mode from SEC to FFF.
- Dilution Control Module (DCM): Each Eclipse FFF channel includes a DCM port which, when combined with the Eclipse DCM module reduces the dilution of sampling eluting from the channel by up to 5x without loss of resolution (up to 10x total range). This feature increases sensitivity and the concentration in collected fractions. It also greatly improves repeatability as a result of the tightly-controlled detector flow rate, as well as the performance of dynamic light scattering measurements.
Interested in more information on the Eclipse?

Mobility
The Mobility system, consisting of the Mobility Module and Mobility channel, combines with the Eclipse to implement electrical/asymmetric-flow FFF (EAF4) in order to measure charge and zeta potential.
Channels
Every Eclipse channel includes a DCM port for increased sensitivity and temperature stabilization for enhanced repeatability. The selection of FFF channels and their applications:

| Channel type | Benefits | Applications |
| Analytical short | Rapid nano/microgram separations | Versatile all-purpose |
| Analytical long | Nano/microgram separations | Polymers, less prone to overloading |
| Semi-preparative | Milligram separations | Extracellular vesicle, virus and LNP isolation |
| Dispersion inlet | For aggregation-prone samples | Monoclonal antibodies, liposomes |
| Mobility EAF4 | Separate by size and charge | Zeta potential distributions |
Pump, autosampler and fraction collector
Eclipse is supplied with industry-standard Agilent 1260 series quaternary pump and autosampler. Other Agilent components are supported, including fraction collector.
VISION™
VISION software is the brains behind FFF. It provides intelligent method design, operation and analysis, with comprehensive control of all FFF components. VISION interfaces with ASTRA – the leading software for characterization of macromolecules and nanoparticles in solution by light scattering.
Interested in learning more about the seamless FFF workflow orchestrated by VISION?

Detectors
DAWN™
The most sensitive MALS detector available, anywhere. Incorporates detectors at 18 angles to determine molar masses from 200 Da to 1 GDa and radii from 10 – 500 nm.
Standard option: ambient temperature
Heated/cooled option: -15 °C to +150 °C
The DAWN offers special options to handle fluorescent samples: fluorescence-blocking filters and an infrared, 785 nm laser. In addition, the DAWN can be combined with dynamic light scattering for even more comprehensive characterization.
Interested in learning more about DAWN?
Online DLS
WyattQELS™ - dynamic light scattering module that can be embedded in the DAWN MALS instrument for on-line DLS.
DynaPro™ NanoStar™ - stand-alone dynamic light scattering detector, may be connected via optical fiber to the DAWN flow cell for on-line DLS.

Optilab™
A unique on-line differential refractometer for measuring concentration of any macromolecule, regardless of chromophores. Temperature controlled from 4 °C to 65 °C. The high-concentration option accommodates protein concentration up to 180 mg/mL.
Interested in learning more about the Optilab?

ASTRA™
Comprehensive software for MALS analysis in chromatography, AF4 or batch mode. ASTRA is available in a 21 CFR Part 11 compliant version and offers additional options such as particle analysis. Provides multiple types of conformation analysis including conformation plots, particle concentration and shape factor.
Interested in learning more about ASTRA?

View this video to learn more about the technology of field-flow fractionation (FFF) implemented in Wyatt’s Eclipse system. Material covered:
- System modules
- Inside the separation channel
- Solvent and sample flow paths
- How Eclipse separates nanoparticles and macromolecules
Learn more about the capabilities of FFF-MALS
Below you will find a partial list of the resources available on our website where you can learn more about FFF-MALS and how it is used by scientists around the world, in academia, industry and government labs for advanced macromolecular and nanoparticle characterization.
Webinars
On-demand webinars presenting the theory and applications of FFF-MALS are available for unrestricted viewing on the FFF-MALS Webinars page. Listed below are some of the most popular:
Biotherapeutics
- Quantification of viral and non-viral vector CQAs
- Vaccines Illuminated: Biophysical characterization, PAT, and quality control via light scattering techniques
- Scientific Town Hall Meeting: Biophysical tools for vaccine characterization and virus research
Nanoparticles
- Characterization of Lipid Nanoparticle Drug Formulations
- Separation and Characterization of Complex (Bio)macromolecular Architectures
- Mass, Size, Composition: Field-Flow Fractionation of Biomolecules and Nanoparticles plus Characterization by Light Scattering and ICP-MS
Polymer R&D
- Structural characterization of functional polymers via light scattering and differential viscometry
- Separation and Characterization of Macromolecules with AF4-MALS
- Structural Characterization of Biopolymers by Analytical Separation Techniques with Advanced Detectors
Protein research and life sciences
Videos
Application Notes
Application notes highlighting the use of FFF-MALS are available for unrestricted viewing on the FFF-MALS Application Notes page. Listed below are some of the most popular:
Biotherapeutics
- WP2606: Exosome characterization with FFF-MALS-DLS
- Aggregate Analyses by Light Scattering - Complementary and Orthogonal
- AN2003: Quantifying AAV aggregation and quality attributes by FFF-MALS
Nanoparticles
- WP2606: Exosome characterization by FFF-MALS-DLS
- WP2608: Lipid Nanoparticle and Liposome Characterization with FFF-MALS-DLS
- WP2611: Characterization of nano-pharmaceuticals with field-flow fractionation and light scattering (FFF-MALS-DLS)
Polymer R&D
- WP1003: Branching revealed: Characterizing molecular structure in synthetic polymers by multi-angle light scattering
- WP2304: Analysis of polymers in organic solvents by FFF-MALS
- WP2303: Lignin and lignosulfonate characterization with SEC-MALS and FFF-MALS
Protein research and life sciences
Theory
Please see the following pages for details of FFF-MALS theory:
Bibliography
An extensive searchable bibliography of publications citing Wyatt FFF-MALS instruments is available at www.wyatt.com/Bibliography. Just open the Advanced Search and click the Multi-Angle Light Scattering box, then enter your search terms below.
Technical Notes
Wyatt offers its customers comprehensive online support via the Wyatt Support Center, including many technical notes that can help make the most of FFF-MALS experiments. If you are a customer and do not have access, please request an account on the Support Log-in page.
At Wyatt, it’s personal
Your success is ours! From the time you purchase a Wyatt instrument, we are there for you every step of the way and give you more than just an instrument warranty. Wyatt provides a full suite of support offerings to help you make the most of your investment. Most importantly, you get unrivaled personal attention to ensure success and productivity.

On-site installation and training
With every instrument purchase, we offer an on-site installation and familiarization visit at your laboratory to ensure proper instrument set-up and function. We also offer customized on-site training tailored to your specific needs. These additional services may be especially desirable if a large group of staff members wants to receive instruction.

First-year service and support
All new Wyatt instruments come with a full year of unlimited telephone and e-mail support as well as our standard full-coverage warranty during the first year. Our congenial staff of Ph.D. scientists and expert technicians provides both technical and application assistance. We will show you how to analyze your data or set up instrument communications by remotely accessing your PC.
After the first year, annual service plans are available for continuing, unlimited telephone support, e-mail support and instrument service. Benefits of the service plans include annual preventive maintenance, on-site calibration, re-qualification, and discounts on parts and labor for repairs. Learn more about service plans.
The specifics of first-year service as well as continuing support may vary by region. Please contact your local Wyatt representative for more information.

IQ/OQ and compliance
Wyatt Technology offers a complete compliance program including documentation and on-site validation for all of its instruments. We provide the resources and tools necessary to ensure compliance. We’ve worked extensively with our partners in compliant environments to build up a robust set of documents and services. Wyatt instruments and software are used in GMP environments at pharmaceutical, biotech, and other regulated industries around the world. Learn more about our Compliance program.

Light Scattering University
Our flagship training program Light Scattering University™ (LSU) is included with every purchase of a light scattering instrument. In the 2, 3 or 4 day course—depending on which system is purchased—LSU students discover advanced data processing methods and alternative analytical tools they may not be familiar with. The students also learn how their MALS and DLS data complements information from other techniques they are using in the lab. Advanced classes are offered for LSU graduates on more detailed techniques and topics.
During LSU our students meet Dr. Philip Wyatt, Founder and Chairman of the Board who gives an acclaimed Light Scattering Instrument Museum tour. Students also meet those who invent, build, service and support the instruments, so they know the team that will support them when back in the lab.
An LSU credit, inclusive of airfare, accommodations and most meals, is included for North American customers who purchase one of our light scattering instruments. While you are here, we work you hard but feed you well—at a variety of Santa Barbara restaurants!
Learn more here.

Service and Support Plans
At Wyatt, we understand that downtime and loss of productivity is critical and have developed comprehensive service offerings — our Platinum, Gold and Silver Service Plans — specifically designed to make preventative maintenance and instrument repair requests quick, smooth and hassle-free. These plans are available on an annual basis after the first-year instrument warranty expires and offer priority phone and e-mail support in addition to instrument service.
The Platinum and Gold Service Plans offer comprehensive on-site preventive maintenance, repair services and software updates. A highly trained Wyatt Field Service Engineer will come to your facility and perform annual or semi-annual preventative maintenance, calibration and optional re-qualification checks. We also offer loaner units should an instrument require factory repair. With our Silver Service Plan, you can expect full instrument calibration and quality control testing, priority service including parts, labor, shipping, as well as the same comprehensive, first-priority technical and application support by phone, email, and screen sharing sessions as offered with the Platinum and Gold Service Plans.
Learn more here.
![]()
Web-based support
Our Customer Support Center contains a wealth of useful resources on everything related to your Wyatt light scattering instruments, software and applications:
- Software updates and bug fixes
- Technical Notes on how to connect and work with your instruments and software
- Video tutorials, software tutorials and educational webinars
- Learning Resource section with New User Guides
- Reference materials such as User's Manuals, Certificates of Analysis for Wyatt-supplied standards, CE and TUV declarations
Register or log in to the Support Center here.
We're Here to Help
Phone number +1 805-681-9009 option 4
Business hours: M-F 8 a.m. - 5 p.m. Pacific
Consulting
By special arrangement, we can provide extended on-site consulting services by an Applications Scientist to set up methods and run samples on your instruments in your lab. This avoids the need to send samples off-site for analysis and allows for intimate, real-time consultations with the best experts available. Availability may be limited to certain geographical regions.
If you would like to have our lab staff analyze samples for you and provide consultations, explore Wyatt's in-house Sample Analysis services.

Wyatt Technology Store
With our online store, world-wide customers can search for parts, consumables and accessories and view product images and part numbers. The store is categorized by Wyatt product families making it easy to know which parts are compatible with your instrument. US and Canadian customers can register for an account and order parts and accessories with either a purchase order or a credit card.
Browse our most popular consumables, review your order history and easily place repeat orders online. If you have questions on which Waters™ Wyatt protein SEC column is best for your application, check out the friendly SEC Columns Guide which will interactively guide you to the recommended column. In addition to ordering parts and accessories, you can also inquire about our training courses, IQ/OQ validation, and service plans.
Visit our online store here.