
Discover Eclipse™
Wyatt Technology’s 6th-generation Eclipse field-flow fractionation (FFF) system represents an entirely new level of FFF technology. It incorporates sophisticated controllers and sensors with built-in intelligence and an intuitive interface to ensure ease of use, reliability and repeatability, suitable for technicians and occasional users as well as expert scientists.
FFF is a versatile technique for achieving analytical and semi-preparative separations. It is applicable to a wide range of analytes including proteins, polymers, viruses, gene vectors, liposomal drug nanoparticles, engineered nanoparticles, colloids and nanoemulsions.
Covering a range of sizes from 1 nm to 10 µm, FFF offers programmable separation power for enhanced resolution within any size range. Downstream of the separation channel, multiple online detectors—from multi-angle and dynamic light scattering (MALS, DLS) to ICP-MS—may be included to analyze complex macromolecular solutions and colloidal suspensions.

Understanding FFF
Asymmetric-flow field-flow fractionation (AF4), implemented in the Eclipse system, is by far the most commonly used FFF separation technique. The AF4 principle is described below.
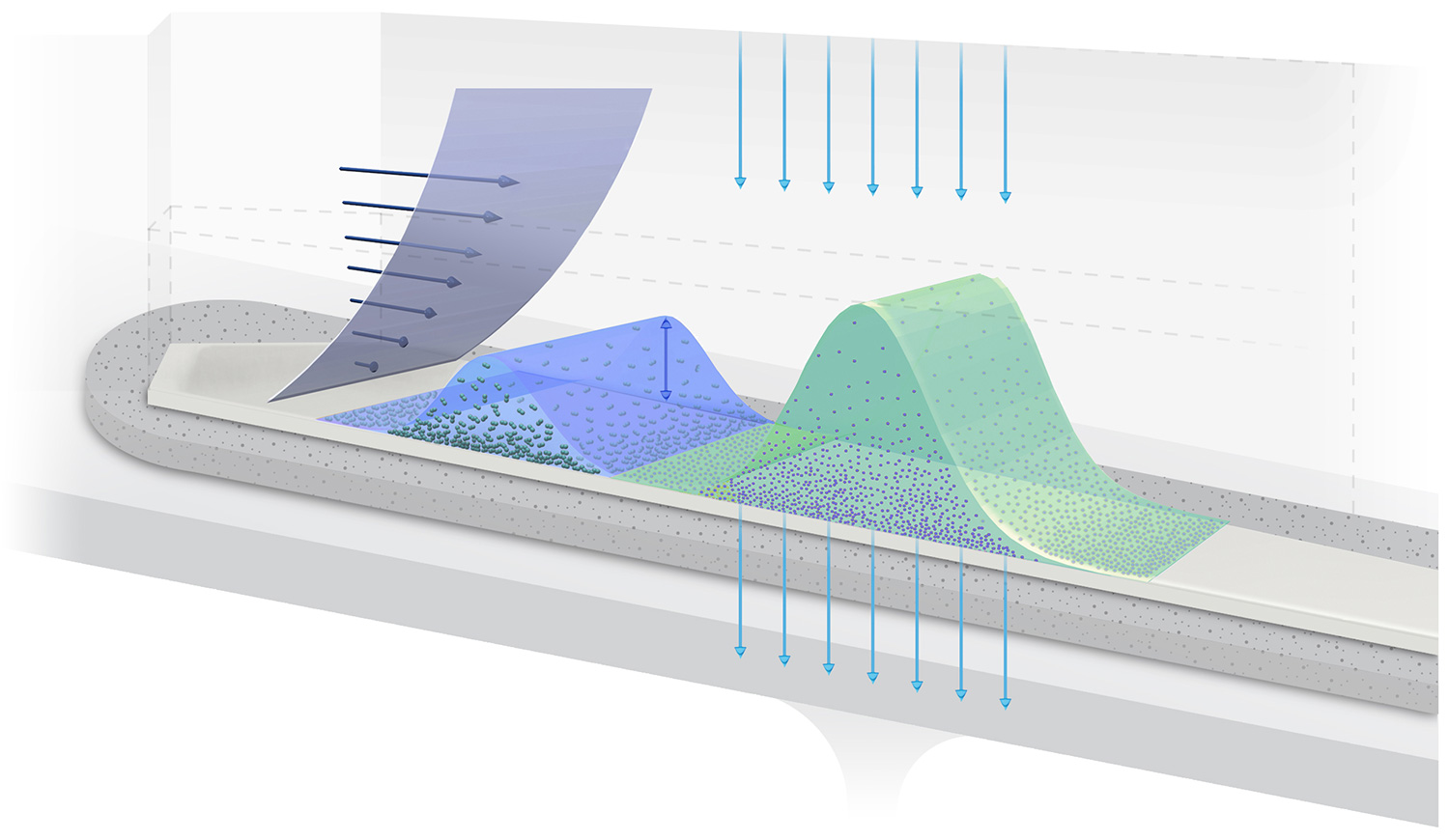
How AF4 separates by size
The AF4 channel
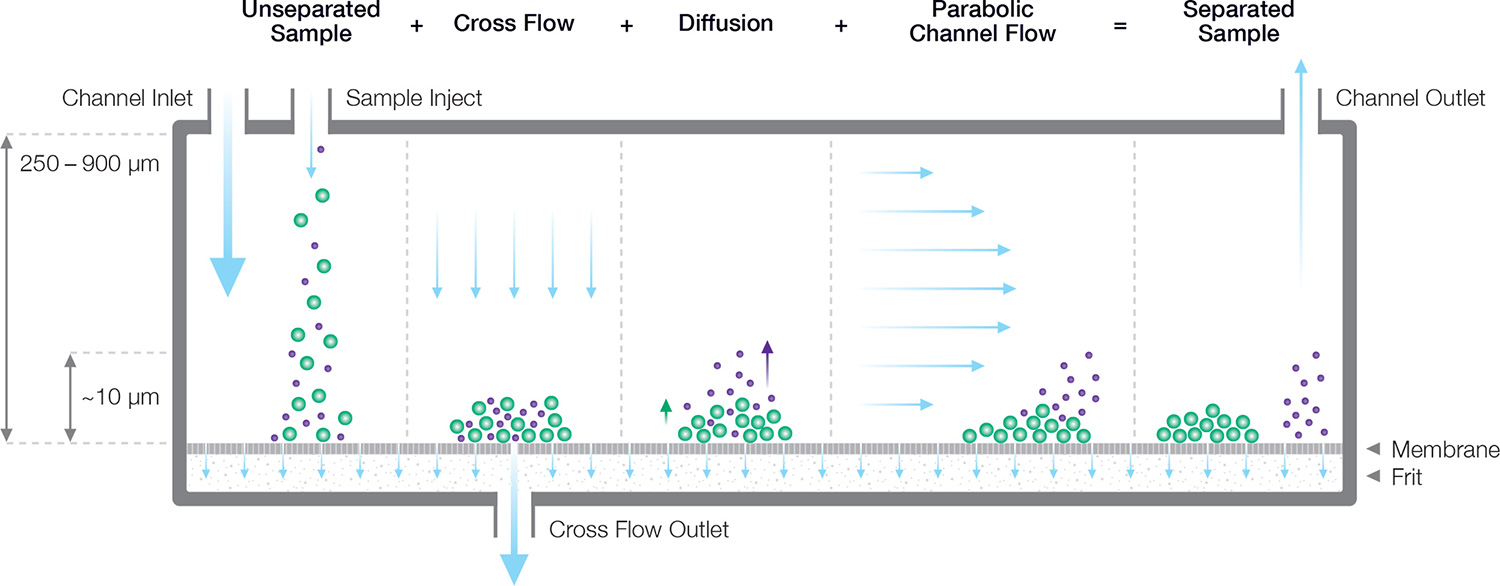
Separation takes place in an open channel consisting of parallel top and bottom plates separated by 200 to 500 µm. Flow within this thin ribbon-like channel is laminar, with a pronounced parabolic flow profile that drives particle separations.
The bottom plate comprises a semi-permeable membrane supported by a frit. The membrane is permeable to solvent but not to the analyte; this essential function is guaranteed by selecting the appropriate membrane pore size, expressed as a molecular-weight cutoff (MWCO) ranging from 1 kDa to 100 kDa.
Balancing forces
The carrier fluid flows parallel to the membrane, but a constriction at the end of the channel forces some of the fluid to pass through the membrane, creating a “cross-flow field” that concentrates the particles towards the membrane. Diffusion acts as a counterforce that drives particles back up into the channel, resulting in a height distribution above the membrane that depends on the particle’s translational diffusion coefficient Dt, and hence hydrodynamic radius Rh, as well as the cross-flow velocity. Separation arises from differential transport velocity in the laminar flow profile according to the particles’ height above the membrane.
Electrical/Asymmetric-Flow FFF
Electrical/asymmetric-flow field-flow fractionation (EAF4) separates by a combination of size and charge. It is similar to AF4, with the addition of electrodes on the top of the channel and below the frit. The shift in retention time arising from applying an electric field enables analysis of the zeta potential of each component in the solution/suspension.
Key Benefits of FFF
Versatile separations with multiple detection options
- High-resolution separation of macromolecules and nanoparticles from 1 nm - 1000 nm and beyond
- No stationary phase - open-channel separation with minimal shear and surface interactions
- Versatile choice of separation channels, permitting sample loads from nanograms to milligrams
- Simply adjust flow ratios to optimize methods, enhance resolution or increase size range, even programmatically during a run
- Online analysis with multi-angle and dynamic light scattering plus UV, RI, fluorescence and/or ICP-MS to determine molar mass, size, conformation, mass or particle concentration, conjugated/encapsulated content or elemental composition
- Collect fractions for on-line analysis (with electron microscopy, MS, NGS, ELISA, etc.)
Macromolecules
Characterize Macromolecules
Eclipse FFF adds to and complements size-exclusion chromatography (SEC) for macromolecular separations. Separation occurs without shear or adverse, non-ideal column interactions, and HMW species are not filtered out or disrupted.
The Eclipse allows you to take full advantage of conventional SEC-MALS and FFF-MALS characterization in one package. The same HPLC pump and autosampler, and the same MALS, DLS, UV and RI detectors are used to characterize macromolecules for molecular weight, size, conformation and conjugation by SEC or FFF. The FFF-SEC switching option lets you switch between the two separation techniques at the click of a mouse.
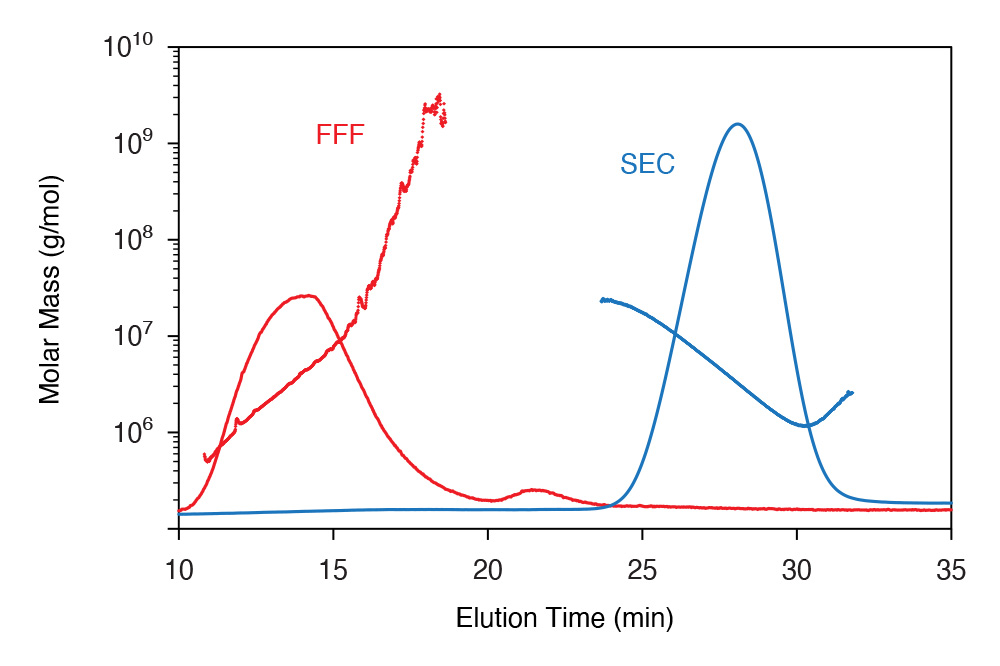
SEC and FFF separations of an extremely heterogeneous and high-molar-mass polysaccharide with molar mass values overlaid on RI signals. SEC is seen to remove the highest HMW fractions while intermediate MW fractions elute non-ideally; FFF separation is nearly ideal with no losses.
Nanoparticles
Characterize Nanoparticles
Common solution-based methods for determining the size and size distributions of nanoparticles, such as batch dynamic light scattering (DLS) and nanoparticle tracking analysis (NTA), cannot offer the resolution, statistical confidence and absolute accuracy of a true separation-based technique that incorporates MALS and DLS downstream detection.
FFF-MALS and FFF-DLS performed with an Eclipse, DAWN® and an embedded WyattQELS™ DLS detection module offer excellent resolution and accuracy in analysis of size distributions. Particle concentration and sizes of viruses, gene vectors and lipid nanoparticles estimated by FFF-MALS match closely TEM-based counts. With additional spectroscopic and RI detectors, encapsulation of nucleic acids or small-molecule drugs can be quantified.
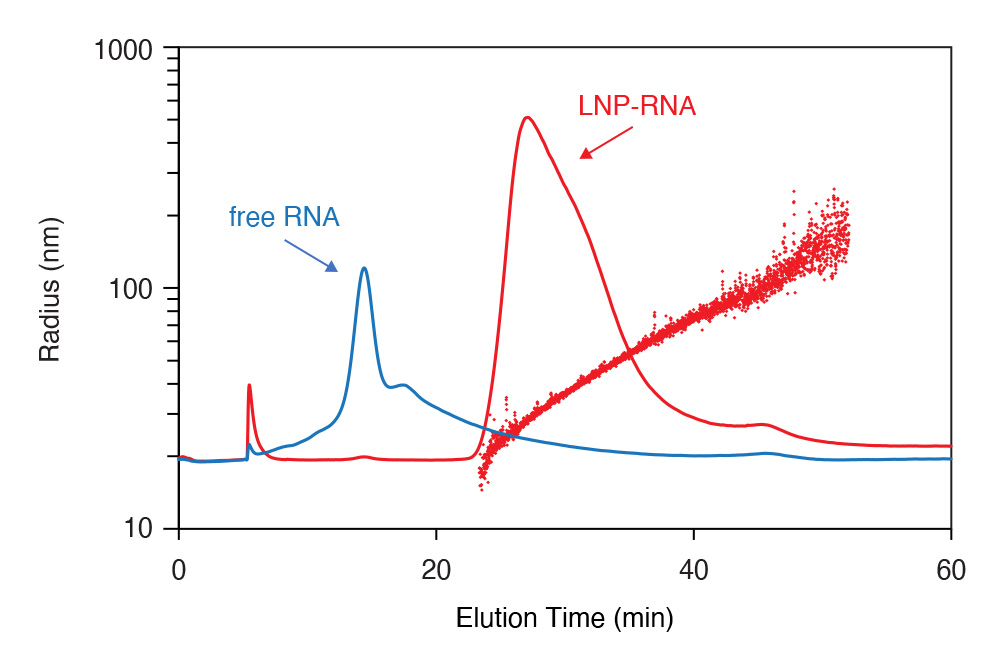
FFF-MALS analysis of lipid nanoparticles for RNA transduction, and free RNA. Very little free RNA was found in the LNP sample indicating complete encapsulation.
Key Benefits of the Eclipse FFF System
- Comprehensive characterization: Integrates with premier online instruments including DAWN™ MALS/DLS instrument, Optilab™ dRI detector, and HPLC UV detector
- Built-in intelligence: Advanced user interface plus sophisticated controllers and sensors let you know if all systems are go
- Computer-aided method design: Eliminates the need for a matrix of sample runs during method development
- Front end and auxiliary modules: Integrates with top-tier products - Waters and industry-standard pump, autosampler, and fraction collector
- Dilution Control Module™ (optional): DCM enhances sensitivity and fraction concentrations by up to 10x; simultaneously, controls the detector flow rate to 0.1% accuracy for highly repeatable methods.
- A channel for every application: Selection of analytical and semi-preparative channels engineered for maximum performance, all DCM-ready
- Guarantee productivity: Gold and Silver Service Plans protect your investment and keep your instruments running at their best

FFF-MALS Products

Wyatt Technology™ offers a complete, turn-key solution for FFF and FFF-MALS-DLS comprising:
Eclipse
The Eclipse FFF instrument incorporates sophisticated flow and pressure controllers to provide robust and repeatable separations. Continuous self-diagnostics and an information-rich, intuitive display provide system status, method progress and alerts should the conditions be sub-optimal.
Options:
- FFF-SEC switching system that automatically transfers the same autosampler, pump and detectors to either FFF or SEC mode
- DCM dilution control module
Mobility
The Mobility EAF4 module controls voltage in the Mobility channel and measures pH and conductivity. In combination with Eclipse and VISION™, Mobility determines zeta potential for each sample component.
DAWN
The most sensitive MALS detector available, anywhere. Incorporates detectors at 18 angles to determine molar masses from 200 Da to 1 GDa and radii from 10 – 500 nm. The WyattQELS™ module adds dynamic light scattering (DLS) capabilities to determines hydrodynamic radii from 0.5 to 300 nm. DLS determines the sizes of macromolecules and nanoparticles as Rh, the hydrodynamic radius, calculated from the diffusion coefficient.
Optilab
A unique on-line differential refractometer for measuring concentration of any macromolecule, regardless of chromophores. The high-concentration Optilab accommodates protein concentration up to 180 mg/mL.
All Wyatt instruments are designed, assembled and tested in the U.S.A.

 Click to enlarge image.
Click to enlarge image.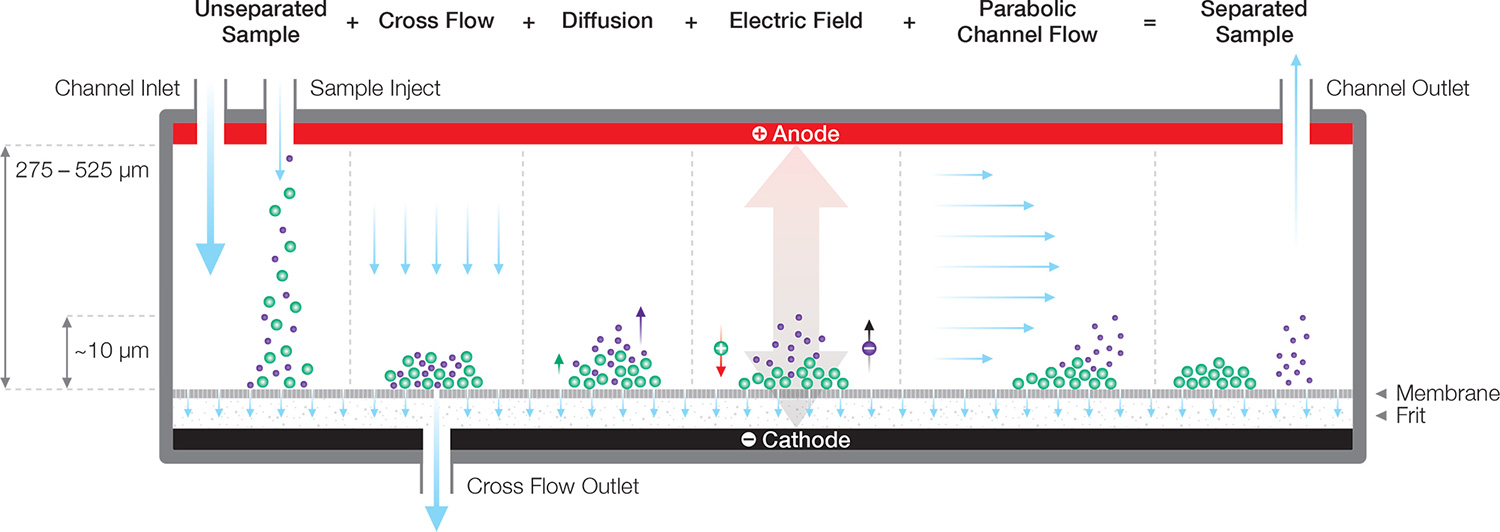 Click to enlarge image.
Click to enlarge image.