Empower™CDS
Software Solutions for MALS
Accelerate high‑quality data generation for proteins, peptides, and other biomolecules with streamlined Empower Software MALS and RI workflows that integrate seamlessly into your existing operations—all within a compliance‑ready, scalable system that delivers optimized, traceable, and reliable data flow.
Features
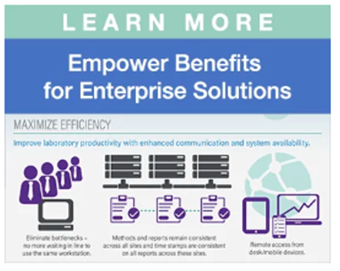
With more than 30 years of proven performance in regulated laboratories worldwide, Empower CDS is trusted to manage data across all types of environments. From single workstations to complex, multi-site global enterprise deployments, Empower Software provides flexible options to scale with your operations.
Designed for today’s quality risk management requirements, Empower Software provides straightforward tools to strengthen data security, ensure traceability, and maintain audit‑ready workflows to enable adherence to compliance with GxP, 21 CFR Part 11, Annex 11, ISO 17025, and other global standards.
- Increase Efficiency: Automate calculations and streamline the complete analytical workflow from acquisition to batch review within a single, unified platform.
- Reduce Risk: Audit trails enable traceable workflows, and automated technical controls minimize manual steps and errors for consistent, reliable data handling across instruments and sites.
- Accelerate Collaboration: Shared methods, centralized data management, controlled user access, and consistent workflows ensure that everyone is working from the same high‑quality data for rapid decision-making.
How it works
Streamlined Workflows. Unmatched Productivity
Empower CDS with integrated MALS acquisition and analysis simplifies complex workflows for both seasoned professionals and new users in regulated environments. Whether you are an expert seeking advanced functionality or a beginner looking for ease of use, Empower Software delivers streamlined performance that keeps your lab operating efficiently and with confidence.

Quality Without Compromise
Strengthen data integrity and minimize risk with advanced SEC‑MALS workflows built directly into Empower CDS for traceable, confident data management in your quality‑critical environments. Assess, control, mitigate risks, and monitor risks throughout the analytical lifecycle within a single, integrated platform supported by automated workflows and technical controls that minimize manual steps, human error, and bias. Leverage Waters experts and the Waters Software Lifecycle Development Audit-Ready Kit to streamline risk-based validation and help your lab stay current and compliant.
Scaled to Your Operations
Harness the full power of your light scattering and chromatographic data with flexible Empower Software Subscriptions and deployments that scale from a single workstation to an enterprise-wide system—on-premises or in the cloud. Unlock streamlined workflows with centralized data management, automation, and compliance across your network for long-term data integrity.

Harmonize With One Software
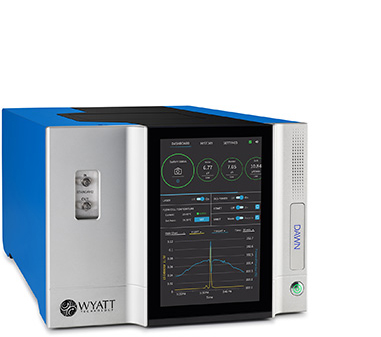
Delivering robust and reliable chromatographic performance, Empower CDS seamlessly integrates with the Waters suite of innovative liquid chromatography systems in addition to other third-party LC, GC, IC, and CE instrumentation. Implementing Empower CDS controlled LC systems with the Waters world-class DAWN MALS instrument line and advanced detectors enables precise multi-angle light scattering analysis for absolute molecular characterization.