UHP-SEC-MALS Analysis of NIST mAb Reference Material
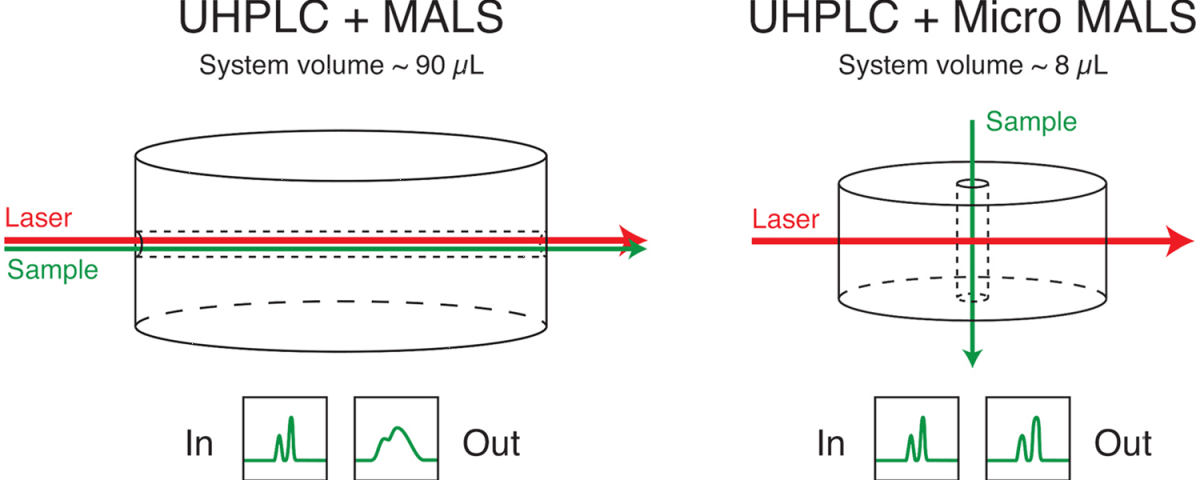
Increasingly UHPLC is being implemented throughout the biopharmaceutical industry, replacing HPLC for characterization of monoclonal antibodies and other therapeutic proteins with reduced consumption of sample and solvent, higher throughput and increased resolution. The microDAWN™ multi-angle light scattering (MALS) detector was developed to meet the need for a low-volume, low-dispersion MALS instrument compatible with size-exclusion UHPLC (SE-UHPLC). UHP-SEC-MALS combines the microDAWN with the microOptilab™ differential refractive index detector and a SE-UHPLC system for absolute molar mass and size measurements of proteins and other macromolecules.
“Measuring proteins with greater speed and resolution while reducing sample size”, authored by Drs. Vincent Hsieh and Philip Wyatt (both of Wyatt Technology™) reports on the application of µSEC-MALS to NIST standard reference material SRM 8671, which was developed as the benchmark molecule against which many biomolecular analytical techniques are assessed and evaluated.
Not only did UHP-SEC-MALS produce data of comparable quality to standard SEC-MALS on monomer and aggregates with far less material and time investment, it was also able to discern and quantify a very small peak representing a fragment–that would appear to be a heavy chain of IgG with molar mass 44.9 kDa–not seen with standard SEC-MALS. An accelerated aggregation course highlighted the ability of the microDAWN to detect and quantify the weight-average molar masses of small high-molecular-weight (oligomer) peaks and resolve minimal amounts of dual-heavy-chain fragments, molar mass of 98.9 kDa, that ride on the monomer peak.
The article appears in Scientific Reports 7: 10030 and may be accessed freely at the nature.com website via DOI:10.1038/s41598-017-09051-1. Congratulations to Drs. Hsieh and Wyatt on this important publication!
