ZoBio Optimizes Solution Behavior of Proteins
ZoBio - February 10, 2016
In many projects, the largest hurdle to be overcome is preparation of a protein that is well-behaved for biophysical and structural biological studies. Often even after achieving high level expression of a soluble, functional variant of the target, it may aggregate under the high concentrations required for many experimental techniques, particularly NMR and X-ray crystallography. However, careful choice of solution conditions can overcome this problem and turn a highly problematic protein into a stable, well-behaved one. How do we find the right solution conditions? With the help of our new SEC-MALS instruments, naturally: the miniDAWN™ and Optilab™. SEC stands for size exclusion chromatography (gel filtration) which, using an ultra-high resolution Superdex Increase column, separates proteins based on size with baseline separation of e.g. a 15 kDa monomer from a 30 kDa dimer. MALS stands for multi-angle light scattering, which together with the Refractive Index detector, allows us to determine the solution size of a protein and the homogeneity of a preparation (also valuable information for QC of protein preparations).
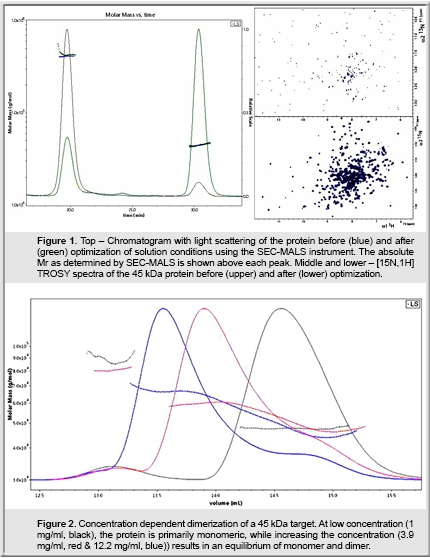
Figure 1 shows data for a 45 kDa protein from a recent project. Although the protein preparation was essentially homogenous by SDS-PAGE, the NMR spectra of the protein were initially very poor. Using the initial conditions, SEC-MALS indicated that 80% of the protein was in a large oligomer of 450 kDa, clearly less than ideal for NMR. By screening different buffers and additives, we could force the equilibrium back to a monomer that is stable for more than a month. Using these conditions throughout the purification also improved the yield of monomer. This stable monomer gives extremely high quality NMR spectra such that we could automatically assign approximately 70% of the backbone resonances in 3 weeks, including acquiring the NMR spectra! The resonance assignment allowed us to rapidly map the binding site of ligands that were elusive in crystallography.
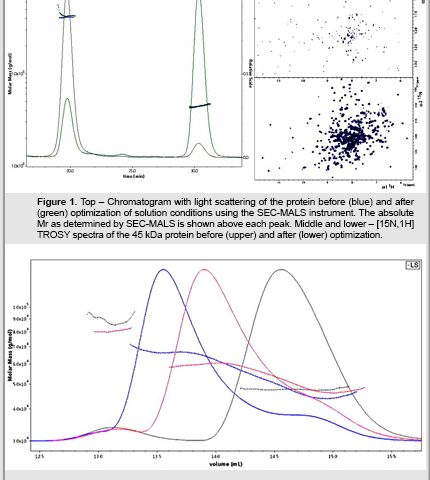
In another example (Figure 2), we used SEC-MALS to demonstrate concentration dependent aggregation of another 45 kDa protein. Again, the protein was homogenous by SDS-PAGE, yet gave very poor NMR spectra. When we looked at the protein in SEC-MALS at the typical concentration one uses for analytical studies (1 mg/ml), the prep was more than 90% monomeric (eluting at 14.7 ml) with a MW of almost exactly 45 kDa across the peak (black trace), with a small component of dimer at 90 kDa (eluting at 13.1 ml). Given the linearity of the Wyatt system, we could follow the behavior of the protein at higher concentrations. At 3.9 mg/ml the prep clearly contains a mix of monomer and dimer that are in rapid exchange with an average MW of about 58 kDa (13.9 ml), as seen in the red trace. At 12.2 mg/ml (blue trace), the equilibrium is further shifted towards dimer with an average MW of 65 kDa. The rapid exchange between monomer and dimer explain the poor quality of the NMR spectrum of this protein. SEC-MALS, particularly when used with our combinatorial biology techniques to sample many thousands of protein variants, can be used to enable your targets for both biophysics and structural biology.