Polymers
Overview
Absolute molar mass & size
Branching & conformation
Co-polymer analysis
Zimm plots
dn/dc measurement
Viscometry & Mark-Houwink
Polymerization kinetics
Pulp & paper products
Polysaccharides
Biodegradation
Application notes
Selected references
The drive to develop synthetic rubber during World War II spurred the first research efforts into theory and practice of light scattering for characterization of macromolecules in solution. Since then light scattering technology has evolved into the sophisticated, robust instruments developed – and continually refined - by Wyatt Technology. Absolute analysis of synthetic polymers to determine molecular weight, size, branching and conformation remains one of the primary applications of multi-angle light scattering.
Sample and reference polymers with the same hydrodynamic volume elute from a GPC column at the same time, but do not have the same molecular weights. SEC-MALS determines polymer molar mass independently of elution time.
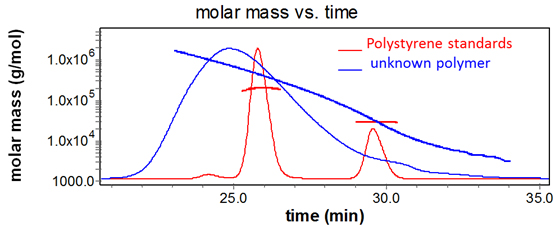
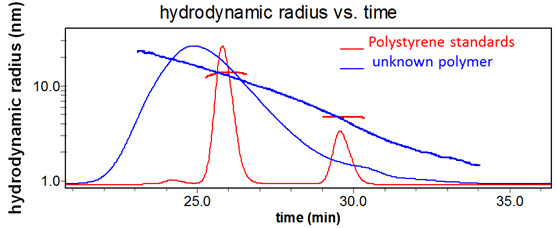
Absolute molar mass & size
 Only multi-angle light scattering (MALS) can determine the absolute molar mass and size distributions of heterogeneous polymers independently of retention time and molecular standards, and regardless of non-ideal column interactions. That's because MALS measures molecular weight and rms radius (a.k.a. radius of gyration) directly from first principles. All you need is a convenient means of size-based separation preceding the on-line MALS detector.
Only multi-angle light scattering (MALS) can determine the absolute molar mass and size distributions of heterogeneous polymers independently of retention time and molecular standards, and regardless of non-ideal column interactions. That's because MALS measures molecular weight and rms radius (a.k.a. radius of gyration) directly from first principles. All you need is a convenient means of size-based separation preceding the on-line MALS detector.
Couple a DAWN™ MALS detector and an Optilab™ refractive index concentration detector to your favorite size exclusion chromatography (SEC) or gel permeation chromatography (GPC) system to create a SEC-MALS absolute characterization tool for polymers. Wyatt detectors interface with HPLC systems from all major vendors.
For more advanced separation capabilities consider the advantages of field-flow fractionation coupled to MALS detectors – FFF-MALS. Wyatt's Eclipse™ FFF system separate nanograms to milligrams over sizes from 1 to 1000 nm, without shear or non-ideal column interactions.
Branching
Design and control of polymer branching permit the creation of synthetic materials with novel mechanical, thermal and rheological properties. The most reliable means of evaluating branching in all types of polymers is multi-angle light scattering, as described by Dr. Stepan Podzimek in 'Branching Revealed: Characterizing Molecular Structure in Synthetic and Natural Polymers by Multi-Angle Light Scattering' (Application Note and On-Demand Webinar).
Branching ratio is determined through the relationship between molar mass and size; both are determined simultaneously and independently via multi-angle light scattering coupled to size exclusion chromatography (SEC-MALS) or field-flow fractionation (FFF-MALS). Molecules smaller than 10 nm in radius require a ViscoStar™ differential viscometer to determine size or else a WyattQELS™ dynamic light scattering module embedded in the MALS detector for size measurements.
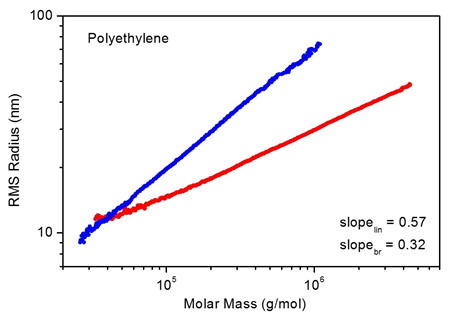
Figure 1. Analysis of branching by comparing conformation plots of linear and branched polystyrene.
While SEC-MALS is suitable for a wide variety of polymers, FFF-MALS using the Eclipse offers many advantages over SEC. FFF-based separation is imperative for certain large, highly branched polymers that elute abnormally from SEC columns.
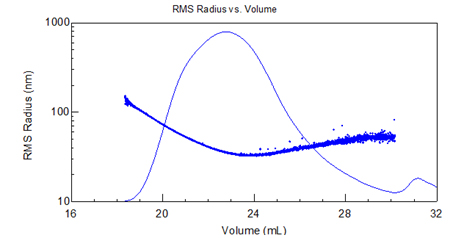
Figure 2. RMS radius vs. elution volume plot of acrylic copolymer. Separation by SEC-MALS exhibits abnormal elution (non-monotonic in molar mass).
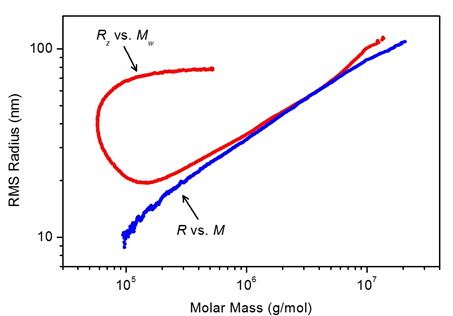
Figure 3. FFF-MALS (plotted in blue) overcomes abnormal elution and anomolous conformation plots exhibited by certain large, highly branched polymers in SEC-MALS (plotted in red).
Conformation
Even if the polymer is not branched, information regarding conformation is available in the relationship between molar mass and size, or between the ratio of rms radius Rg to hydrodynamic radius Rh.
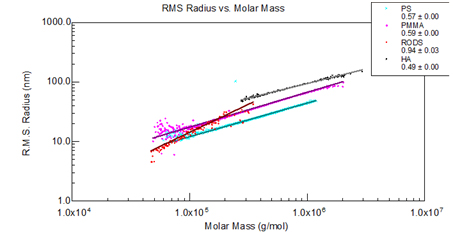
Figure 4. Polystyrene, PMMA, cellulosic rods and hyaluronic acid exhibit different slopes corresponding to different conformations.
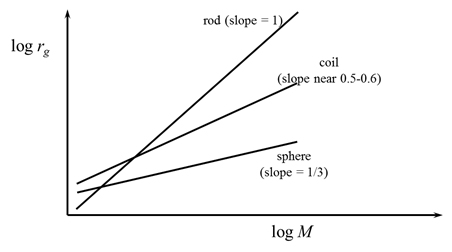
Figure 5. Theoretical relationship between mass and radius for different conformations.
Copolymer Analysis
Co-polymers are not generally amenable to standard GPC analysis because no reference standards are available to represent elution time vs. molar mass. Even with standard SEC-MALS it is difficult or even impossible to analyze heterogeneous distributions of co-polymers, since in general dn/dc – a property that depends on the chemical composition but not the structure of the polymer, and a requisite parameter in MALS analysis – varies across the chromatogram unpredictably.
However, it is possible to carry out co-polymer analysis via the triple-detection system: SEC + UV-MALS-RI, or in some cases SEC + NIR-MALS-RI, if the two polymer components exhibit significantly different responses to the two concentration signals. In this scenario, the ASTRA™ software acquires concentration data from two distinct concentration detectors (ultraviolet UV and refractive index RI, or near-infrared NIR and RI). RI is measured via an Optilab differential refractometer, MALS is measured via a DAWN multi-angle light scattering detector, and UV or NIR via third-party online detectors.
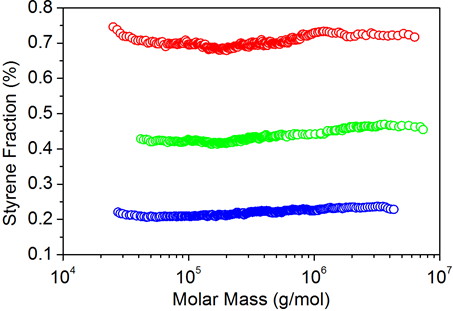
Figure 1. Plots of styrene fraction versus molar mass for copolymers of styrene-butyl acrylate prepared by emulsion polymerization. The feed styrene fraction: 23%, 48% and 73%.
Given known polymer responses to each of the two concentration detection signals it is possible to calculate the ratio of concentrations of the two; this information is combined with the MALS signal to determine the molar mass of each polymer in the complex. Hence at each elution time this analysis provides the overall molar mass as well as the total molar masses of each constituent in the co-polymer.
Unfractionated Measurements: Zimm Plots
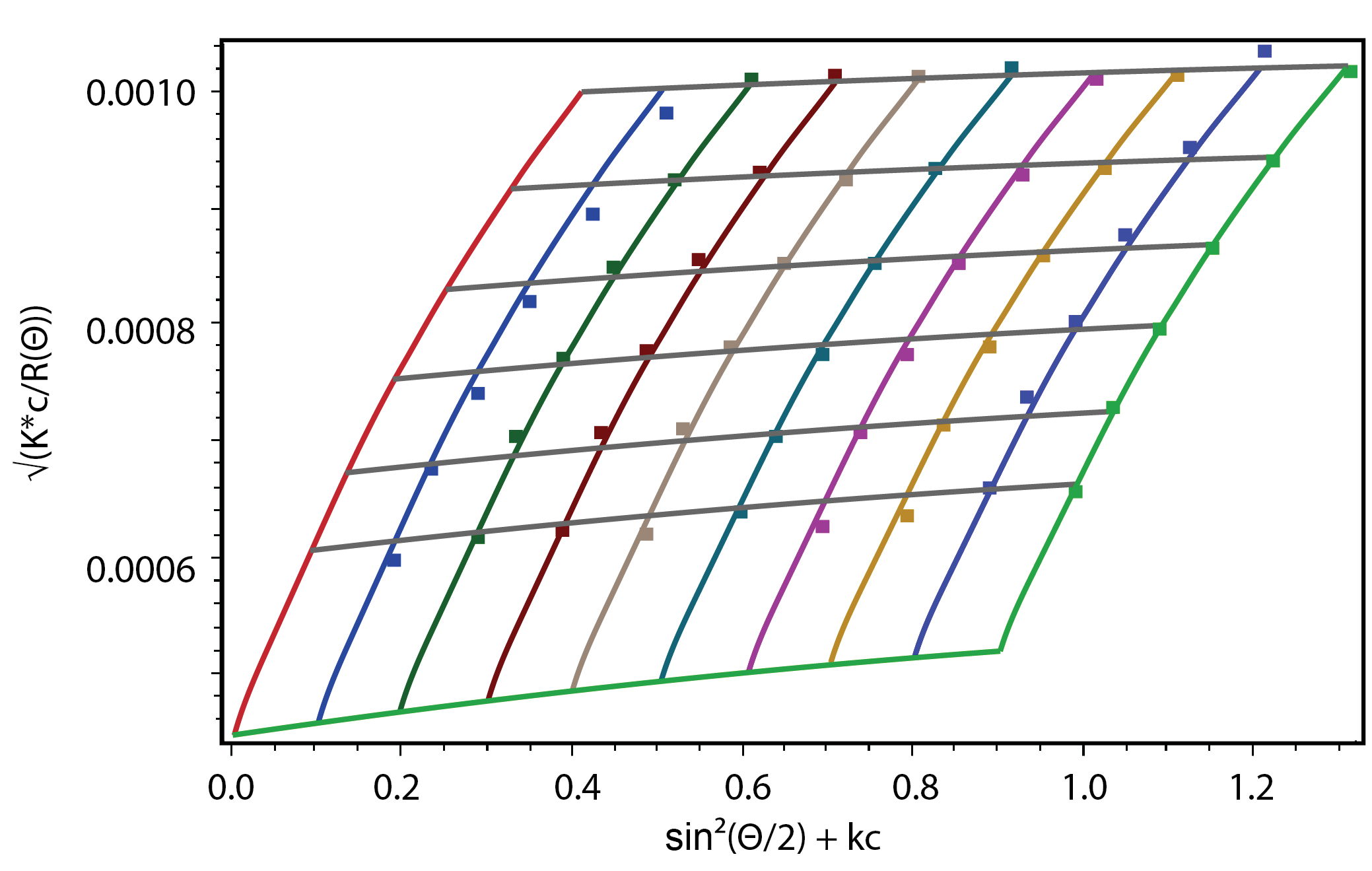 Some polymers are too fragile to run through a gel permeation chromatography (GPC) column without degradation, e.g., a macroligand which can dissociate from labile metals. Others, such as large PMMA molecules, may be too large for GPC. These materials can still be characterized by means of batch MALS, which determines weight-average molecular weight Mw and Z-averaged rms radius rg,z. Accurate determination of these values requires a 'Zimm plot' analysis, i.e., a measurement of light scattering intensity as a function of angle and concentration, without separation.
Some polymers are too fragile to run through a gel permeation chromatography (GPC) column without degradation, e.g., a macroligand which can dissociate from labile metals. Others, such as large PMMA molecules, may be too large for GPC. These materials can still be characterized by means of batch MALS, which determines weight-average molecular weight Mw and Z-averaged rms radius rg,z. Accurate determination of these values requires a 'Zimm plot' analysis, i.e., a measurement of light scattering intensity as a function of angle and concentration, without separation.
The traditional method for making Zimm plots involves manual preparation of a series of aliquots with increasing concentrations. Batch MALS measurements are made in a scintillation vial using the Batch Conversion Kit. Alternatively, in the microbatch method the aliquots are injected into the MALS flow cell. In either case ASTRA software analyzes the data via a global fit of all angles and concentrations to a single light scattering equation. The results include Mw, rg,z and the second virial coefficient A2 which indicates solute-solute and solute-solvent interactions.
Several convenient methods for making Zimm plots utilize automation to create a series of dilutions from a single stock solution, injecting the samples into the MALS and RI flow cells, acquiring the concentration and light scattering data automatically and calculating the three parameters. This automation may be carried out with the Calypso™ composition-gradient system or by programming an autosampler with a large injection loop.
Differential Refractive Index Determination
The differential refractive index dn/dc quantifies the change of refractive index of a solution with concentration of the analyte. There are at least three reasons for measuring dn/dc of a polymer sample:
- In order to use a differential refractometer to measure concentration online with GPC – dn/dc represents the instrument response
- Analysis of MALS data requires knowledge of dn/dc at the MALS wavelength
- The fraction of each monomer in a co-polymer solution can be estimated if the dn/dc values of the two monomers differ significantly
Manual measurements of dn/dc are readily carried out on the Optilab differential refractometer by means of the WISH™ Injection Module. Automated measurements may be carried out by combining the Optilab with a Calypso composition-gradient system.
SEC-IV and Universal Calibration
SEC-IV utilizes intrinsic viscosity and empirically determined Mark-Houwink parameters to determine polymer molar masses. The Mark-Houwink parameters depend on the polymer, solvent and conformation.
Though not as rigorous as SEC-MALS, Universal Calibration (UC) is commonly utilized in the analysis of molecular weights of linear polymers. UC determines hydrodynamic volume from retention time on the column, and calculates molar mass at each elution volume from the ratio of hydrodynamic volume and intrinsic viscosity (this analysis is less accurate than SEC-MALS because it ignores the possibility of non-ideal sample-column interactions, and requires reference standards compatible with the mobile phase).
The ViscoStar on-line differential viscometer and Optilab differential refractometer operate in tandem along with ASTRA software to analyze molar mass and intrinsic viscosity distributions of a polymer sample by means of SEC-IV or UC.
These techniques are particularly useful when the refactive indices of the polymer and solvent are closely matched, making the polymer essentially invisible since it will not scatter appreciable quantities of light (dn/dc~0).
Measuring Mark-Houwink parameters
The relationship between molar mass and intrinsic viscosity for a given type of polymer, indicative of molecular conformation, is described by the Mark-Houwink parameter. This value may be obtained in two ways:
- by purely viscometric means, using UC to characterize both molar mass and intrinsic viscosity.
- by SEC-MALS-IV-RI wherein molar mass is determined by the combination of a DAWN MALS detector and Optilab, while intrinsic viscosity is determined with a ViscoStar and Optilab. The analysis is carried out in the ASTRA software.
Kinetics of Reactions and Degradation
Since light scattering intensity is directly proportional to molar mass, it is an excellent means of monitoring the progression of a polymerization reaction. The angular dependence of multi-angle light scattering indicates size for additional diagnostics, and dynamic light scattering may be included for further characterization.
When reaction time scales are significantly longer than the time required to perform a separation (typically 30 minutes), the ideal means of characterizing the polymerization process is regular withdrawal of aliquots from the reaction vessel and analysis by means of SEC-MALS. Reactions over shorter times must necessarily be evaluated in batch (unfractionated) mode which provides average molar masses and sizes rather than full distributions. The Calypso composition-gradient system is a convenient means of preparing, mixing and injecting multi-component solutions into a MALS system such as the DAWN.
Wood, pulp and paper products
Some interesting analytical challenges are posed by biopolymers from wood pulp and similar natural sources.
Lignins
Lignins tend to absorb laser light and fluoresce. Both phenomena lead to erroneous measurements of molar mass and size, unless the instrumentation and software can overcome these sources of error. The DAWN offers an optional infrared laser plus narrow bandpass optical filters on the detectors to minimize both total fluorescence and the amount reaching the detectors. In addition, the 'Forward Monitor' detector in the DAWN measures laser absorption by the sample to allow the ASTRA software to correct the measured molar mass.
Cellulose
Cellulose forms large, rod-shaped nanocrystals. While they may not be accommodated by standard GPC, these are readily separated by the Eclipse field-flow fractionation system, then measured downstream with a DAWN and possibly the WyattQELS integrated dynamic light scattering module. The shape of the nanocrystals is assessed from the dependence of molar mass on size, then fed back into the analysis to refine the accuracy of mass and size distributions.
Heparin
An important component of the blood coagulation chain, heparin requires careful, complete characterization. Light scattering offers several methods in which to do so:
- SEC-MALS with a DAWN multi-angle light scattering detector and Optilab differential refractive index detector, for absolute molar mass and size distributions
- Automated electrophoretic light scattering with a Mobius, to assess charged impurities such as super-sulfated heparin (chondroitin sulfate)
- Composition-gradient MALS with a Calypso and DAWN, to study binding of proteins to heparin
Hydroxyethyl starch (HES)
HES is a polysaccharide-based blood replacement manufactured from corn and potato raw materials. The two sources result in HES with different conformations arising from differing degrees of branching. SEC-MALS not only determines accurate molecular weight independent of conformation, it also quantifies the conformation and branching ratio for a deeper understanding of the two HES processes. As described by Karyakin et al., SEC-MALS is an essential component of HES quality control.
Biodegradation
Light scattering is an excellent means for assessing the degradation of biopolymers as a result of exposure to heat, light, high or low pH, and other stimuli.
Aggregation and fragmentation
Some common forms of biodegradation are aggregation and fragmentation, both readily characterized with high information content via SEC-MALS. Simply take aliquots of a biopolymer sample before and after exposure to environmental or chemical stress and inject onto an appropriate GPC column followed by MALS and dRI detection. The analysis can provide several means of quantifying degradation, e.g. shifts in Mn, Mw and Mz, representing the number-, weight- and z-averaged molecular weights of the sample, respectively. Biodegradation may also lead to changes in molecular conformation, indicated in SEC-MALS by the ratio of rms radius Rg to molecular weight.
Particle formation
Another manifestation of biodegradation is the formation or dissociation of particulates. For a quick assessment of nanoparticle populations, DLS is ideal, requiring little sample and very little time for preparation an measurement. While the size of monodisperse particles can be measured accurately by DLS, size distributions tend to be more qualitative than quantitative. A more thorough analysis is provided by FFF-MALS, which separates both soluble and insoluble components with excellent resolution so they may be analyzed by downstream light scattering detectors.
Kinetics
While very slow processes are amenable to analysis by SEC-MALS via periodic sampling from a reaction vessel, batch MALS, batch DLS and CG-MALS offer alternative approaches to analyzing kinetics of more rapid reactions.
If there is enough time between initiating the reaction, and mixing and pipetting to a cuvette, batch MALS or batch DLS can work. The Calypso can perform this automatically with a dead time of just a few seconds.
Application Notes
WP1003: Branching revealed: Characterizing molecular structure in synthetic polymers by multi-angle light scattering
This white paper provides a brief overview of the basic principles of the detection and characterization of polymer branching by means of multi-angle light scattering. SEC-MALS is most appropriate for small and intermediate polymers, while AF4-MALS provides superior separation and characterization of large and highly branched macromolecules.
WP2303: Lignin and lignosulfonate characterization with SEC-MALS and FFF-MALS
Lignin and lignosulfonate analysis are among the most demanding applications of SEC-MALS and FFF-MALS. This white paper highlights the successful characterization of their absolute molar mass distributions via FFF-MALS, aided by the advanced technical features of the DAWN and the Eclipse.
AN1301: Characterization of PLGA using SEC-MALS-IV
Branching is used to adjust the pharmaco-kinetic, biodegradable and mechanical properties of PLGA. Characterization of branched PLGA by conventional size-exclusion chromatography (SEC) suffers from the lack of suitable calibration standards. In this application note, PLGA samples were analyzed by SEC-MALS-IV to determine absolute molar mass distributions and conformations, with no need for column calibration.
AN1302: Characterization of hyaluronic acid with online multi-angle light scattering and differential viscometry
The physiochemical behavior of hyaluronic acid (HA) is tied closely to material characteristics such as the weight-average molecular weight, polydispersity, intrinsic viscosity, and molecular conformation. This application note explains how differential viscometry is used in combination with SEC-MALS to analyze HA from a variety of sources, yielding detailed understanding of these properties and why sample-averaged literature values are so different.
WP1004: Absolute characterization of polymers with light scattering and UHP‐SEC
UHP‐SEC offers multiple benefits for synthetic polymer characterization, but is more sensitive to column calibration errors and drift than traditional HP‐SEC. This white paper demonstrates how UHP-SEC‐MALS accurately and reliably determines the molecular weight, size and conformation of polymers ranging from hundreds to millions of g/mol, while maintaining high resolution and sensitivity and low solvent consumption.
AN1005: Identifying short-chain branched polymers with conformational analysis
This application notes describes how to differentiate between long- and short-chain branching, and characterize short-chain branching using a silicone polymer example.
Selected References
Abbas, S.; Lodge, T. P. Depletion interactions: effects of added homopolymer on ordered phases formed by spherical block copolymer micelles. Macromolecules 2008, 41, 8895-8902.
Bielawski, C. W.; Benitez, D.; Grubbs, R. H. An "endless" route to cyclic polymers. Science 2002, 297, 2041-2044.
Podzimek, S. Importance of multi-angle light scattering in polyolefin characterization. Macromol. Symp. 2013, 330, 81-91.
Striegel, A. M. Influence of chain architecture on the mechanochemical degradation of macromolecules. J. Biochem. Bioph. Meth. 2003, 56, 117-139.
Striegel, A. M., Pitkanen L. Detection orthogonality in macromolecular separations: role of the on‑Line viscometer in characterizing polymers at conditions of “spectroscopic invisibility”. Chromatographia 2015, 78, 743-751.
Tarazona, M. P; Saiz, E. Combination of SEC/MALS experimental procedures and theoretical analysis for studying the solution properties of macromolecules. J. Biochem. Bioph. Meth. 2003, 56, 95-116.
