What are some considerations and strategies when working with fluorescing samples?

Introduction
Fluorescent macromolecules pose an interesting challenge for analytical methods based on light scattering as the light emitted by the samples themselves can be detected as if it were scattered light. This excess detected light can impact the resulting data. For static light scattering techniques like multi-angle light scattering (MALS), this can lead to increased signal strength that leads to incorrect molar mass values. For dynamic light scattering (DLS), some sample fluorescence can be tolerated because DLS measures intensity fluctuations from Brownian motion and does not depend on absolute light scattering intensity; however, the fluorescence background may eventually dominate the signal, leading to high count rates and noisy signals. Furthermore, if the excitation wavelength is close to the DLS instrument laser wavelength, you may not obtain a useable signal.
So, what are some strategies for overcoming fluorescence? The first step is understanding the level and nature of fluorescence at your instrument’s laser wavelength. (In last month’s Ask the Expert we showed some ways to identify if a sample is absorbing and fluorescing in the wavelength of your instrument’s laser.) There are two main strategies for addressing sample fluorescence in light scattering:
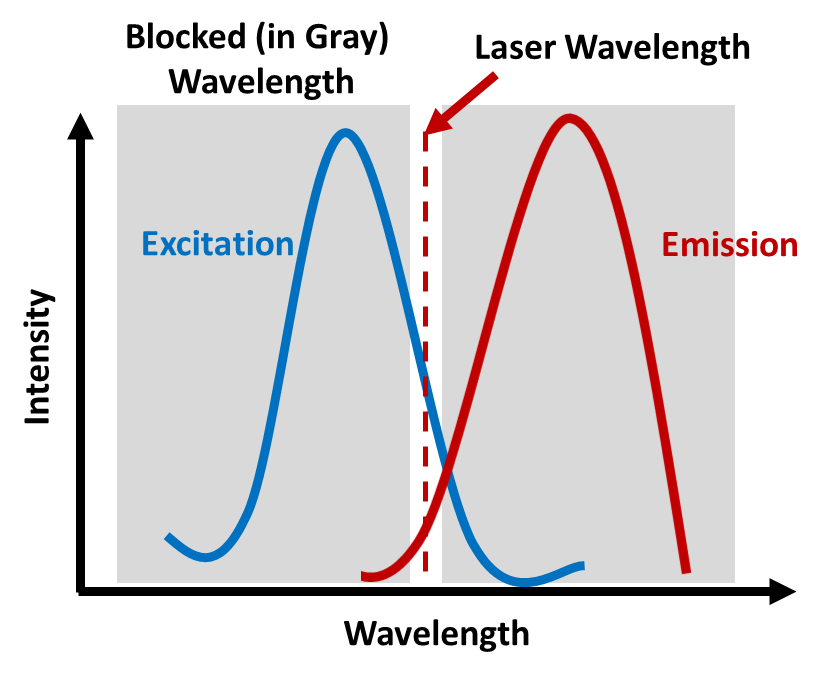
- Installing narrow bandwidth interference filters so that only the scattered light near the laser wavelength of the detector will reach photodiode detectors.
- Utilizing a laser with an incident wavelength that does not overlap with the sample’s excitation spectrum.
Interference filters
The default MALS laser wavelength for the DAWN®, miniDAWN®, and microDAWN™ is around 658 nm, which for many samples completely avoids the excitation band. In situations where 658 nm lies within the excitation spectrum, an understanding of the emission spectrum can help determine an appropriate strategy. If the sample’s emission spectrum differs by more than 10 nm from the laser wavelength, then fluorescence-blocking filters, which have a relatively wide passband of 20 nm, can be used. These operate by blocking all wavelengths except for the laser wavelength ± 10 nm, for example, allowing only 658 ± 10 nm light through and blocking all other light as illustrated.
785-nm laser option
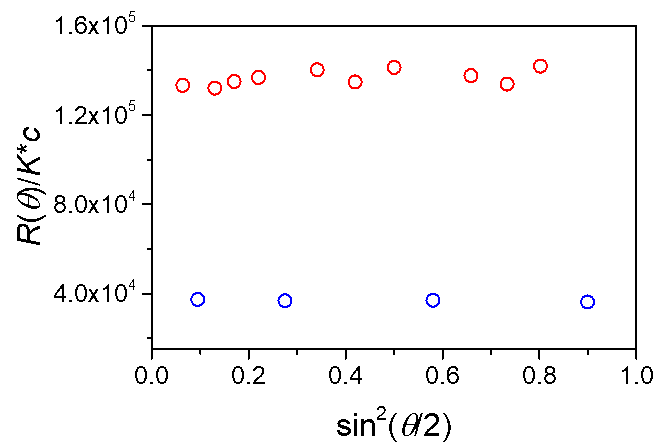
Interference filters are an option for the DAWN that can be installed in the field any time after purchase. The chart here shows the signals of photodiodes covered with interference filters (blue) and without interference filters (red).
While interference filters prevent the fluorescing light being attributed to the sample scattering intensity, they don’t account for absorption by the sample. To check for absorption and correct for it, see last month’s Ask the Expert.
If the fluorescence is strong near the laser wavelength, interference filters may not be sufficient. During the manufacturing of a DAWN instrument, the laser can be configured to have either a 658-nm or 785-nm laser option. The longer 785-nm laser wavelength may altogether avoid excitation wavelengths. The main drawback of the near-IR laser is that the sensitivity is roughly a third compared to the traditional 658-nm laser. Interference filters at 785 nm with a 20 nm bandpass are also available. In all cases with fluorescence-blocking interference filters, it is recommended to install them in at least three angles for the DAWN instrument depending on the size of the sample. Additionally, for accurate dn/dc values, we also offer an Optilab® RI detector at a wavelength-matched 785 nm for such systems.
DLS measurements
For DLS measurements, a number of options are available when fluorescence affects the autocorrelation function (ACF). The DynaPro® NanoStar® is available with a standard 660-nm or custom 785-nm laser, with the near-IR laser less prone to excite fluorescence. The DynaPro Plate Reader uses an 830-nm laser, where fewer fluorescent labels or polymers will emit; however, these higher wavelengths come at the cost of less sensitivity for a given laser power. The Mobius™ is available with a standard 532-nm laser but has the benefit of being compatible with an inline narrow passband fluorescence-blocking filter on the avalanche photodiode (APD).
Conclusion
In some cases, fluorescence is too strong even under near-IR laser wavelengths with fluorescence-blocking filters. In these cases, differential viscometry measurements with the ViscoStar® may provide the optimal means for characterizing fluorescent samples, as intrinsic viscosity can be used to estimate molar mass via the Mark Houwink-Sakurada analysis and universal calibration, which is more robust than traditional column calibration, and also allows for determination of the hydrodynamic radius.
We’ll discuss strategies for using light scattering techniques with fluorescing samples in our next Ask the Expert. Do you have a question? Contact our experts here in Customer Support. We’re happy to help! Call +1 (805) 681-9009 option 4.