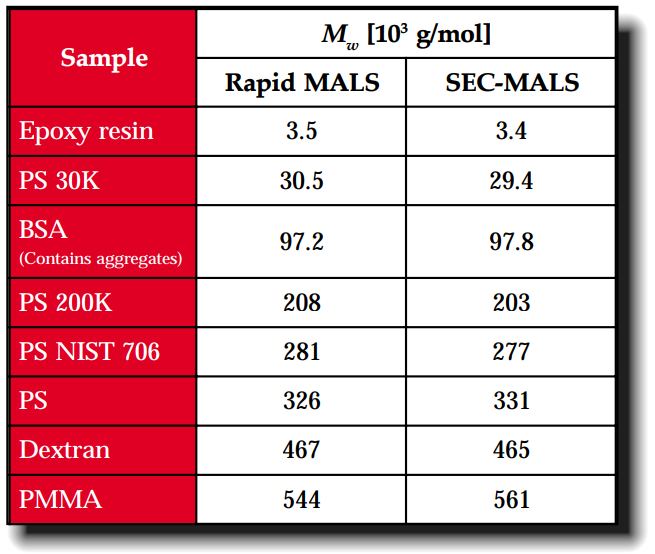
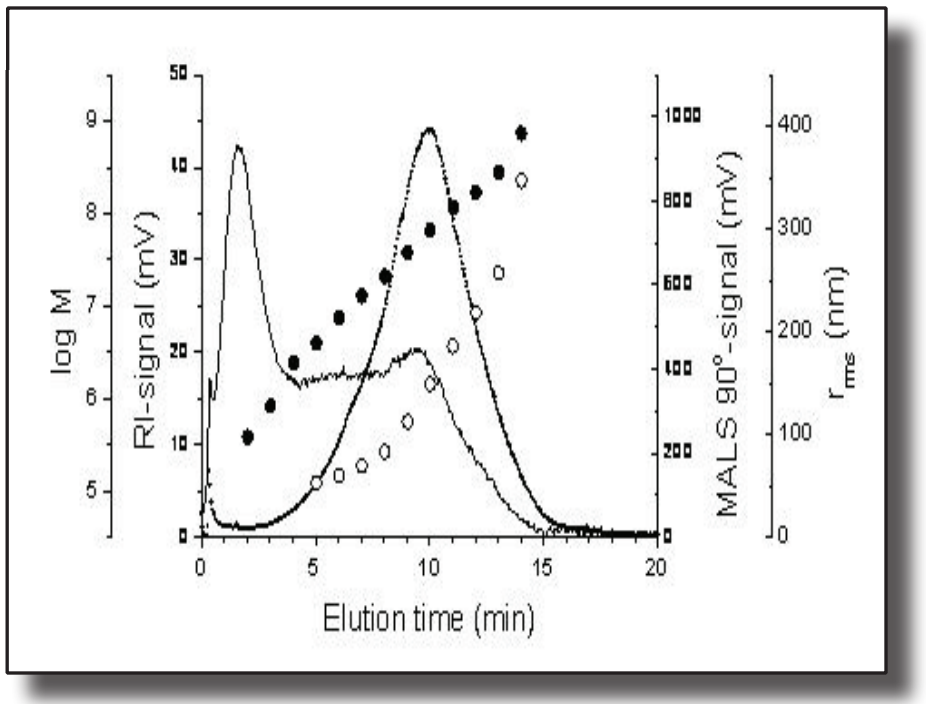
This application note presents results obtained using an experimental set-up consisting of an HPLC pump, a sample injector, a multi-angle light scattering (MALS) photometer, a differential refractive index (DRI) detector, and an SEC guard column.
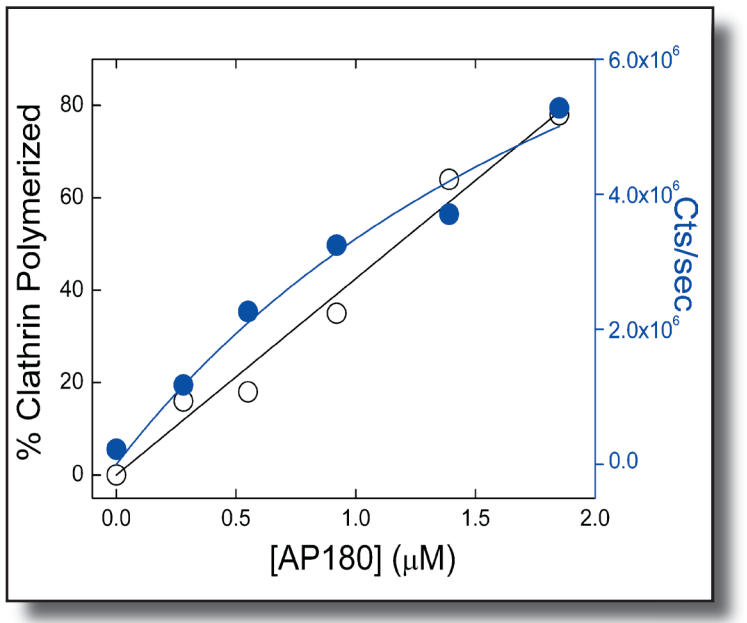
We have developed a novel assay using a DynaPro Dynamic Light Scattering (DLS) instrument to study the dynamics of clathrin coated vesicle (CCV) assembly and disassembly. Learn more by reading our application note.
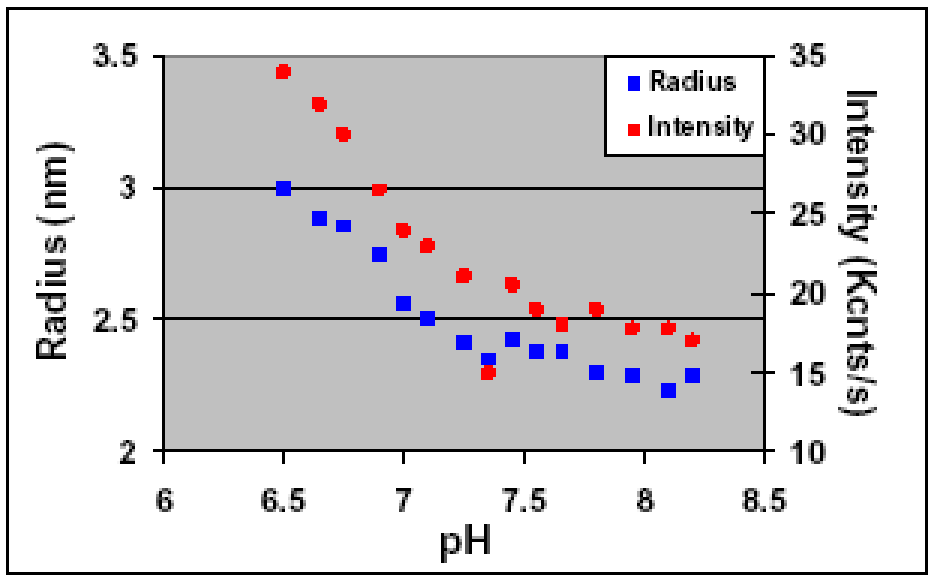
In this experiment, a DynaPro light scattering instrument was used to monitor the protein stability and homogeneity in different buffers. Learn more by reading our application note.
Glycinamide ribonucleotide transformylase (GART) catalyzes formyl group transfer in the de novo purine biosynthetic pathway in cells. Because of the dependence of cancer cell growth on purine, the chemo-therapeutic inhibition of GART and other transformylase enzymes is a potential method for combating cancer. Learn more in our application note.
Surfactants are routinely incorporated into protein formulations to 1) solubilize hydrophobic proteins or 2) to act as chaotropic agents against protein aggregation. The sample examined in this study is a partially unfolded derivative of a parent compound that is currently in clinical trials as a treatment for neurological disorders. Learn more by reading our application note.
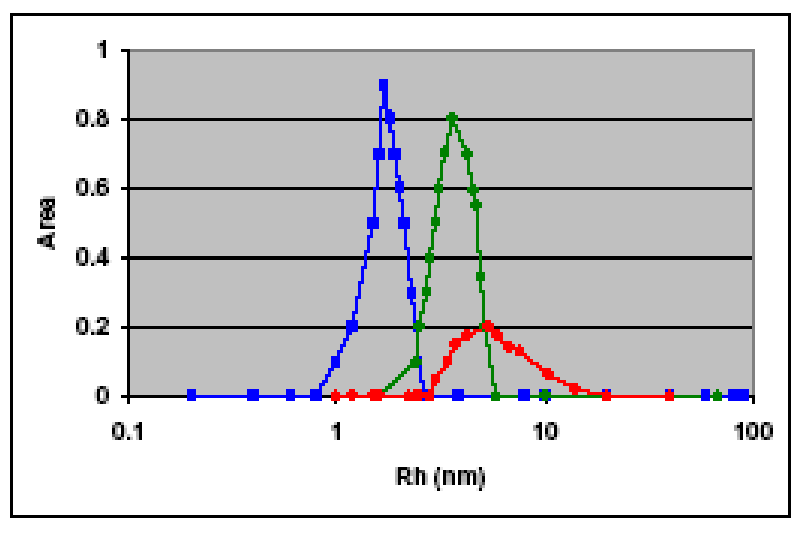
Because of its sensitivity to particle size and polydispersity, dynamic light scattering is a valuable tool for characterizing vesicle suspensions. Learn more in our application note.
The interfacing of nanoparticles (NPs) and biology is a paradigm shift for non-viral medical gene therapeutics. Using a DynaPro DLS system, the formation of a NP-gene construct formed from 13 nm gold (AuNP) and a 1900 base pair gene which encodes for dsRed express fluorescent protein can be systematically evaluated for different gene assembly strategies onto the surface of a AuNP.
The detailed in situ analysis of inorganic and organometallic clusters is becoming increasingly important as researchers now routinely synthesize cluster compounds for use as secondary building units. In the series of experiments which follow, we used DLS to study the formation and reactivity of the well-characterized zirconium tetramer, as well as a recently characterized zirconium-glycine cluster.
This application note proves that AF4 is a more efficient separation technique for branched polymers than traditionally-used SEC. In contrast to SEC-MALS, A4FMALS provides linear conformation plots with no virtual upswings, which allow accurate characterization of branched polymers. Learn more.
The molar mass distribution of starches has been conventionally determined by SEC. However, materials with large molecular weights, such as amylopectins, elute in the void volume, may be partially retained in the column as well as suffer degradation due to shearing forces. Learn more in our application note.
High-performance size-exclusion chromatography (HPSEC) is a traditional structural assay and was employed annually to monitor the integrity of antitoxin treatment for botulism, but recently health agencies have raised the concern of aggregation. Multi-Angle Light Scattering (MALS) of antitoxin separated by asymmetric field-flow fractionation (AFFFMALS) was developed as orthogonal assays to our HPSEC. Learn more by reading our application note.
Recent reports by the FDA and the European Union acknowledge the lack of rugged analysis and charac- terization methods for nanomaterials as a major limit- ing factor to the final establishment of nanotechnologies. We present here results indicating that AF4-MALS-FD can become the unparalleled technique for the analysis and optical char- acterization of new, structured, fluorescent nanomaterials. Learn more.