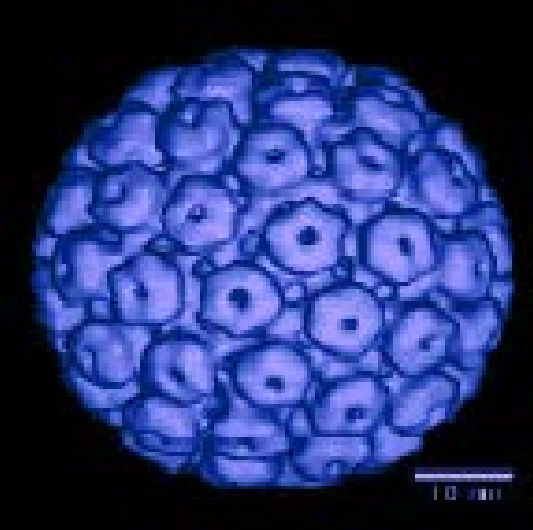
Hyaluronic acid's (HA) distinctive viscoelastic properties, coupled with its biocompatibility and resorbability have led to a wide range of biomedical and pharmaceutical applications. Learn more by viewing our scientific poster.
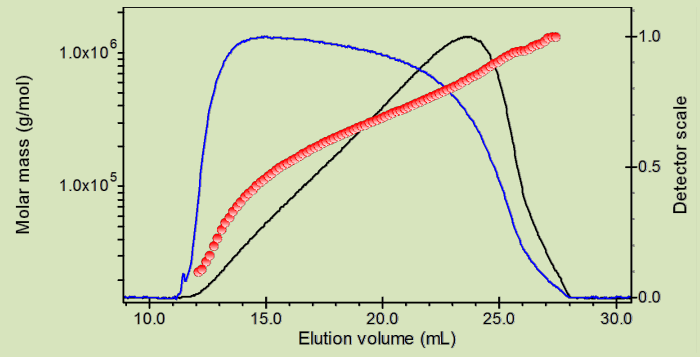
To gain deeper insight into the enzymatic structures, the team used Size-exclusion chromatography (SEC), Multi-Angle Light Scattering (MALS), UV detection and DRI detection. Learn more by viewing our scientific poster.
Concerns about potential removal of large aggregates by the SEC column as well as sample alteration due to dilution and solvent exchange call for methods orthogonal to SEC to detect and quantify aggregates present in a protein solution. In this scientific poster, we discuss the necessity and benefits of using orthogonal light scattering approaches through a case study of two different lots of Pierce BSA. Learn more.
Cellulose ether samples can be used for evaluation of the SEC fractionation of high MW samples having the same chemical compositions. Learn more by viewing our scientific poster.
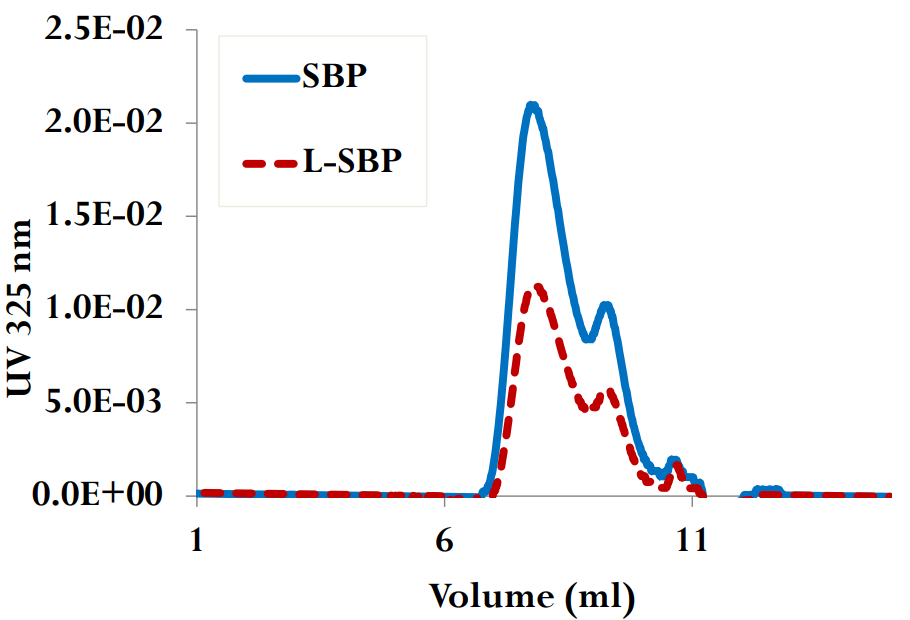
Learn more about our findings on sugar beet pectin's effectiveness as an emulsifying agent over a wide range of pH, ionic strength, heat, proteolysis and other process conditions by viewing our scientific poster.
The Eclipse, an Asymmetric-Flow Field Flow Fractionation apparatus, in line with a set of detectors, can provide simultaneous characterization of key traits of VLP. Learn more by viewing our scientific poster.
Asymmetrical flow field-flow fractionation (AF4) provides high-resolution separations for characterizing a wide range of macromolecules and colloidal particles. This method has been called the most generally useful of any of the field-flow fractionation (FFF) family. Learn more by viewing our scientific poster.
Liposomes and lipid nanoparticles serve as versatile drug delivery systems, offering control over composition, structure, and morphology, where these properties can be tailored to each specific pharmaceutical application. Comprehensive characterization of the particles by FFF-MALS-DLS is essential for rapid and effective pharmaceutical product development as well as for regulatory approval.
This white paper gives a summary of how the Wyatt Eclipse® Field-Flow Fractionation system coupled to MALS and DLS allows for size-based isolation plus detailed and comprehensive characterization of exosomes.
Size-exclusion chromatography is commonly used to separate and analyze proteins and other biomacromolecules. However, in order to reliably determine their basic biophysical properties in solution, an absolute, independent means of characterization must be added downstream of the separation step.
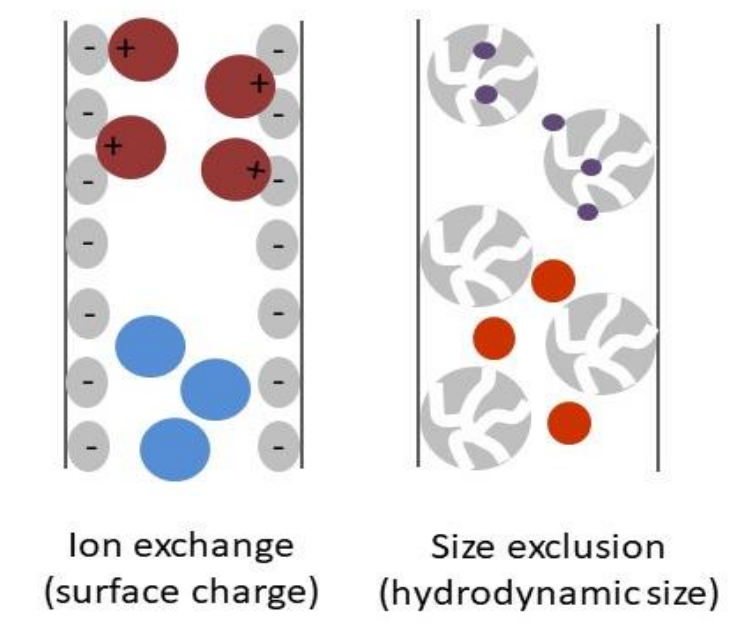
Ion-exchange chromatography coupled to multi-angle light scattering (IEX-MALS) allows absolute molar mass determination of proteins, protein complexes and peptides in a heterogenous sample. Read more here.
Perhaps one of the most versatile sets of nanoparticle characterization tools is based on light scattering, utilizing the three basic flavors of analytical light scattering: multi-angle, dynamic and electrophoretic. Auxiliary techniques that enhance and complement light scattering include field-flow fractionation (FFF), which separates nanoparticles according to size prior to analysis, and inductively-coupled plasma mass spectrometry (ICP-MS) for analysis of elemental composition. Read more here.